Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)
Debanjana Adhikari posted an Question
- CSIR NET
- Physical Sciences
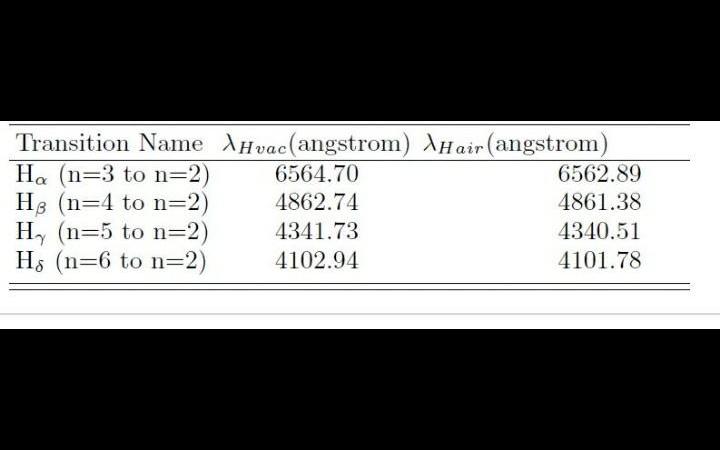
1.11. a sample of ordinary hydrogen (ih) gas in a discharge tube was seen to emit the usual balmar spectrum. on careful examina- tion, however, it was found tha
1.11. A sample of ordinary hydrogen (iH) gas in a discharge tube was seen to emit the usual Balmar spectrum. On careful examina- tion, however, it was found that the H. line in the spectrum was spit into two fine lines, one an intense line at 656.28 nm, and the other a faint line at 656.04 nm. From this, one can conclude that jahir.iitb@gmail.com physicsguide.in/net Sk Jahiruddin Atomic Molecular: June 2020 the gas sample had a small impurity of [TIFR 2015] (a) H (b) H (c) H (d) H20
- 0 Likes
- 6 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Dhairya sharma
first three wave length of isotope , hydrogen , deutrium and tritium are respectively 656.28 , 656.10 , 656.04
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Abhishek singh
Option A is correct. Because H alpha line of Balmer series of Tritium(option A) is 656.04 nm. Hence, the impurity is Tritium.
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
 >
>

 >
>









Chandra prakash
see attached file it's very helpful for you dear.. it's simple method to find this type of prob.