Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shivam Sharma posted an Question
- CSIR NET
- Earth Sciences
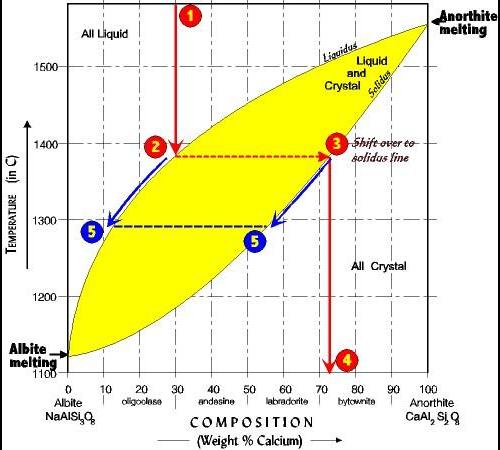
Fig. 1 and fig. 2 given below represent schematic phase relations in two binary systems at identical and constant pressure. a and b have eutectic relationship w
Fig. 1 and Fig. 2 given below represent schematic phase relations in two binary systems at identical and constant pressure. A and B have eutectic relationship while C and D form a solid solution. The temperature scale (Y-axis) is same in both figures. R1, R2, R3 and R4 represent four rock compositions in these two systems. Write the sequence in which melts will start forming in these rocks (RI, R2, R3 and R4) with increasing temperature (without changing pressure). Explain with justification, the order in which the solid component will completely melt (under equilibrium condition) in R1, R2, R3 and R4 as heating proceeds. Liquid Liquid A R1 R2 B C R3 R4 D Fig. Fig. 2
- 0 Likes
- 3 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Rahul kumar Best Answer
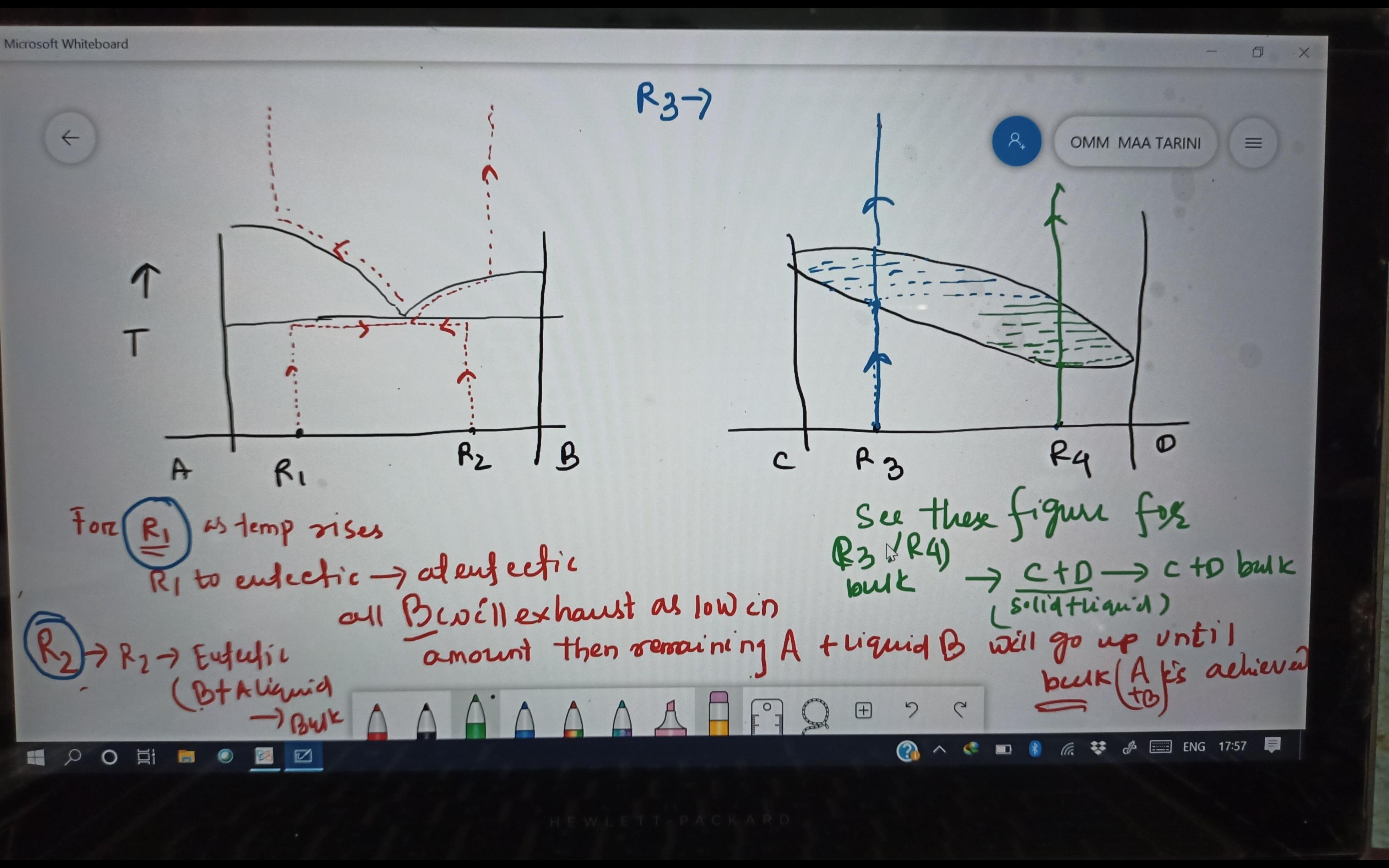
R1: Since R1 is a part of eutectic system, the first melt will be of eutectic composition (mixture of A and B) at eutectic temperature. As soon as composition B will be over in rock, the temperature will increase and melting will produce only composition A now. R2: Since R2 is a part of eutectic system, the first melt will be of eutectic composition (mixture of A and B) at eutectic temperature. As soon as composition A will be over in rock, the temperature will increase and melting will produce only composition B now. R3: The first melting will occur at solidus temp (directly above point R3); the composition of melt will be the horizontal projection of solidus curve intersecting liquids. The melt composition will enrich in composition C and when everything will be turned into melt phase (at liquidus temperature right above R3); the temperature will only increase and the only available phase will be liquid. R4: The first melting will occur at solidus temp (directly above point R4); the composition of melt will be the horizontal projection of solidus curve intersecting liquids. The melt composition will enrich in composition C and when everything will be turned into melt phase (at liquidus temperature right above R4); the temperature will only increase and the only available phase will be liquid. For details, refer to the attached figure which indicates the changing composition of liquid (melt) and solid (rock) with temperature.
![cropped776993321340344345.jpg]()
![eduncle-logo-app]()
So the sequence in which melt form is R1 then R2 and so on ?
![eduncle-logo-app]()
Yes, R1, R2.... are rock compositions and with increasing temperature the sequence of melt composition is written in the answer that I have already sent.
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>

 >
>








Rahul kumar
Yes, R1, R2.... are rock compositions and with increasing temperature the sequence of melt composition is written in the answer that I have already sent.