Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)
Debanjana Adhikari posted an Question
- CSIR NET
- Physical Sciences
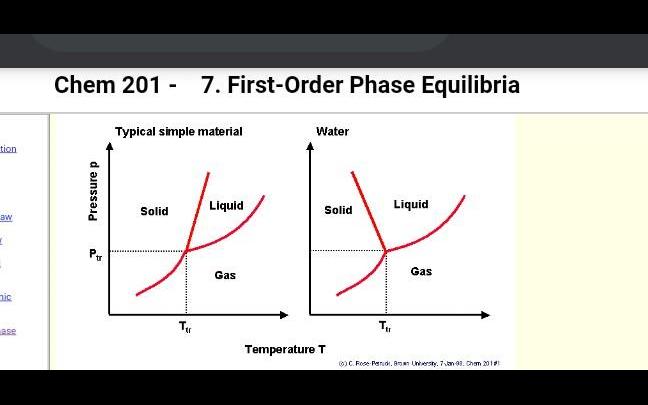
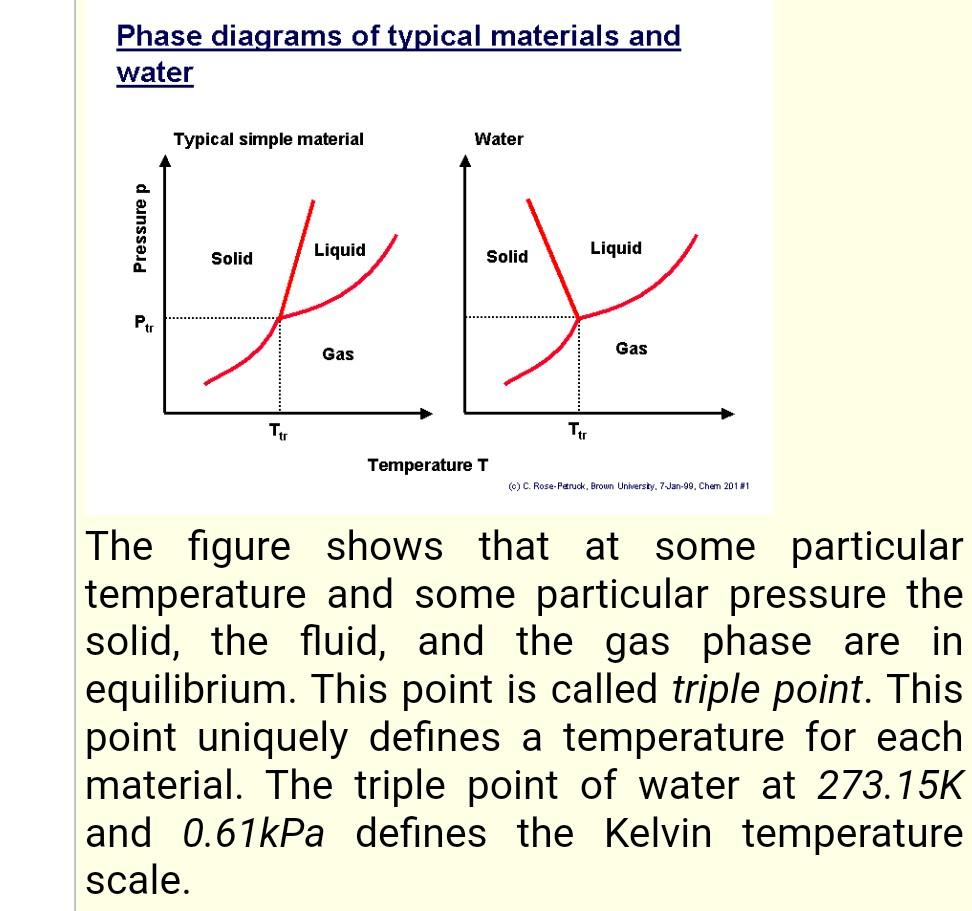
Identify which one is a first order phase tran- sition? (a) a liquid to gas transition at its critical temperature (b) a liquid to gas transition close to its t
Identify which one is a first order phase tran- sition? (a) A liquid to gas transition at its critical temperature (b) A liquid to gas transition close to its triple point (c) A paramagnetic to ferromagnetic transi- tion in the absence of s magnetic field (d) A metal to superconductor transition in the absence of s magnetic field
- 0 Likes
- 5 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Chandra prakash
clearly option (c )and (d ) are not first order phase transition as it involves symmetry variation . so now confusion can arise only with option (a) and (b ) . in order to avoid that confusion, the key point is the definition of critical point. beyond the critical point, no phase transition is possible, therefore it cannot be right answer...... hence, the correct ans option is ( b) .............
-
![comment-profile-img]() >
>
Dhairya sharma
First-order phase transitions are those that involve a latent heat. During such a transition, a system either absorbs or releases a fixed (and typically large) amount of energy per volume. ... Second-order phase transitions are also called "continuous phase transitions". so b is correct.
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
 >
>
 >
>

 >
>









Chandra prakash
I hope my explanation... u understood