Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)- IIT JAM
- Chemistry (CY)
123. forthe followingrcacton, co, +h,0 h,co, the entropy change (4swas calculated to be-96 jk mo'. the enthalpychange (ah) was measured to be-45 k" mol". thus r
123. Forthe followingrcacton, CO, +H,0 H,Co, the entropy change (4Swas calculated to be-96 JK mo'. The enthalpychange (AH) was measured to be-45 K" mol". Thus reaction is expected to be a spontaneous process. the total change in entropy ensrundings (a) +54 JKmo c)-45096 JK° mol (b)-96JK mo (d)-44O04 JK mo
- 0 Likes
- 2 Comments
- 0 Shares
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>

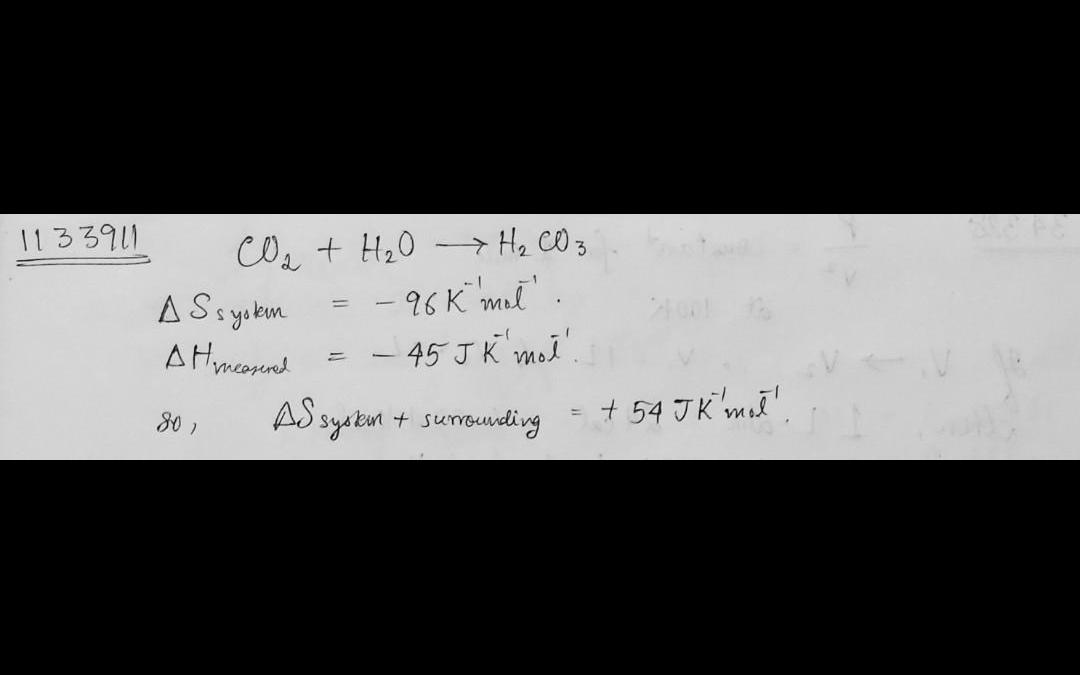







Muskan 1
i am unable to understand by this this is very direct