Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)
Imdadul Haque posted an Question
- IIT JAM
- Chemistry (CY)
+ 2. (a) for the complex mn(h,o), ion, the mean pairing energy p) is found to be 28800 cm. the magnitude of 10 dq or a, is 2 1000 cm. calculate the crystal ficl
+ 2. (a) For the complex Mn(H,O), ion, the mean pairing energy P) is found to be 28800 cm. The magnitude of 10 Dq or A, is 2 1000 cm. Calculate the Crystal Ficld Stabilisation Energy (CFSE) to high spin and low spin state. Which state is more stable? 1
- 0 Likes
- 3 Comments
- 0 Shares
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>

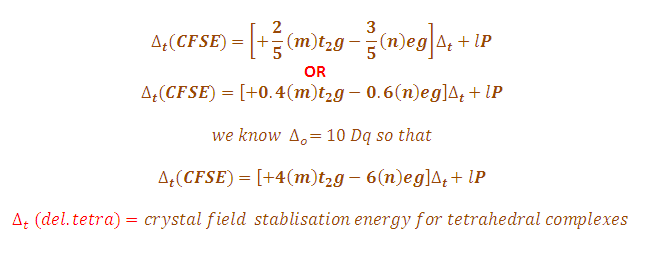
 >
>
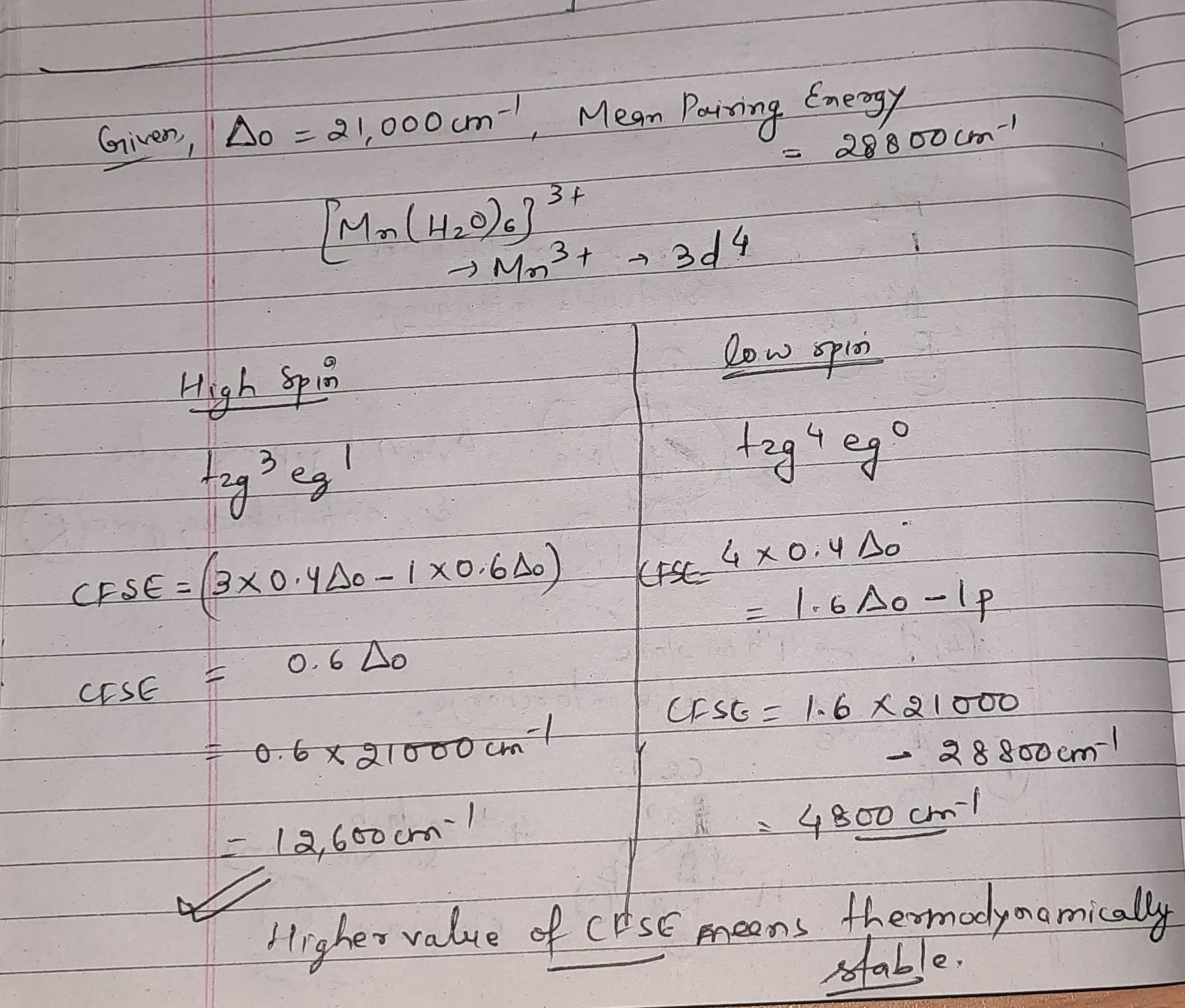








Imdadul haque
Thank You so much Sir