Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)- IIT JAM
- Chemistry (CY)
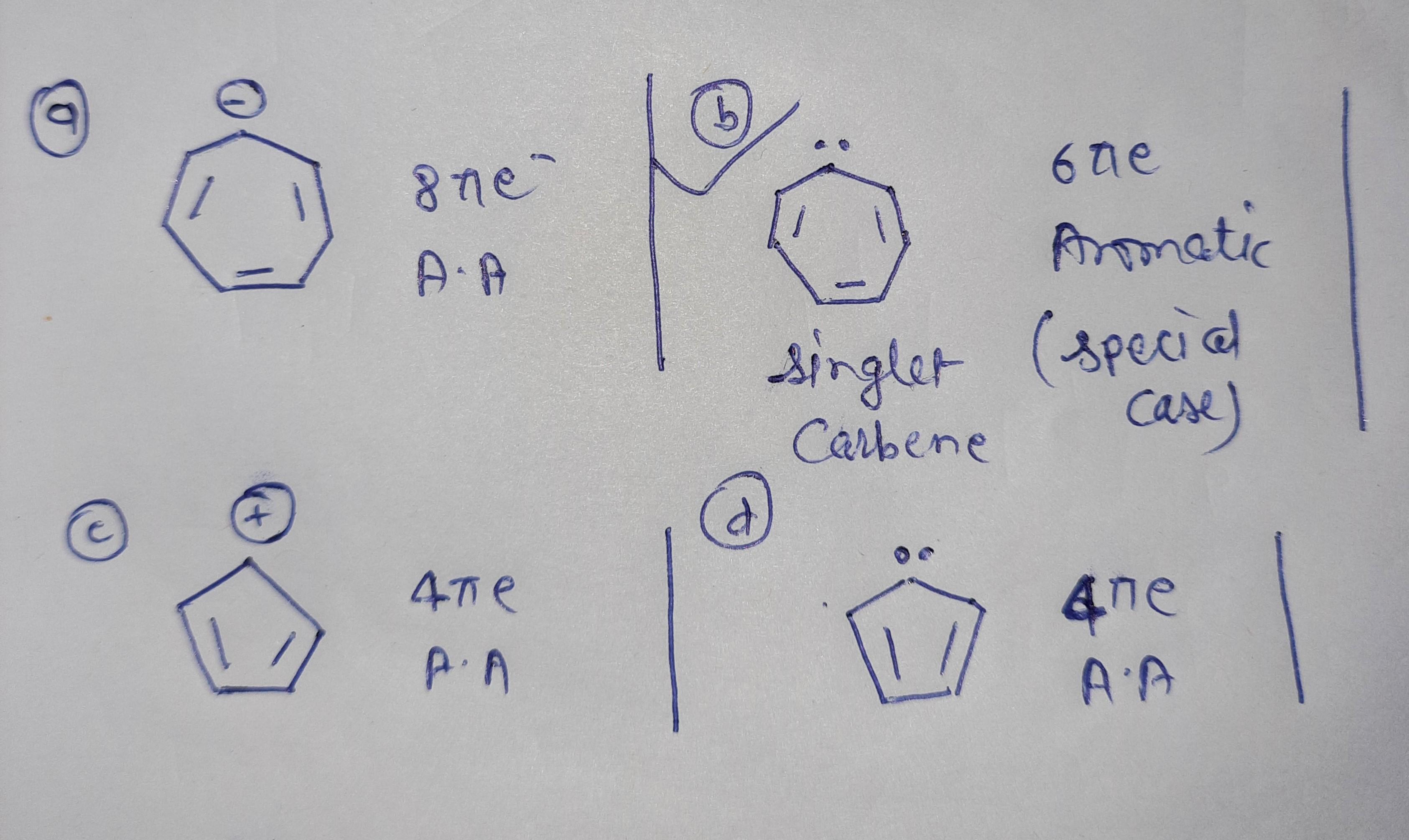
37 goc please ans my following questions as soon as possible
- 0 Likes
- 3 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Priyanshu kumar
![best-answer]()
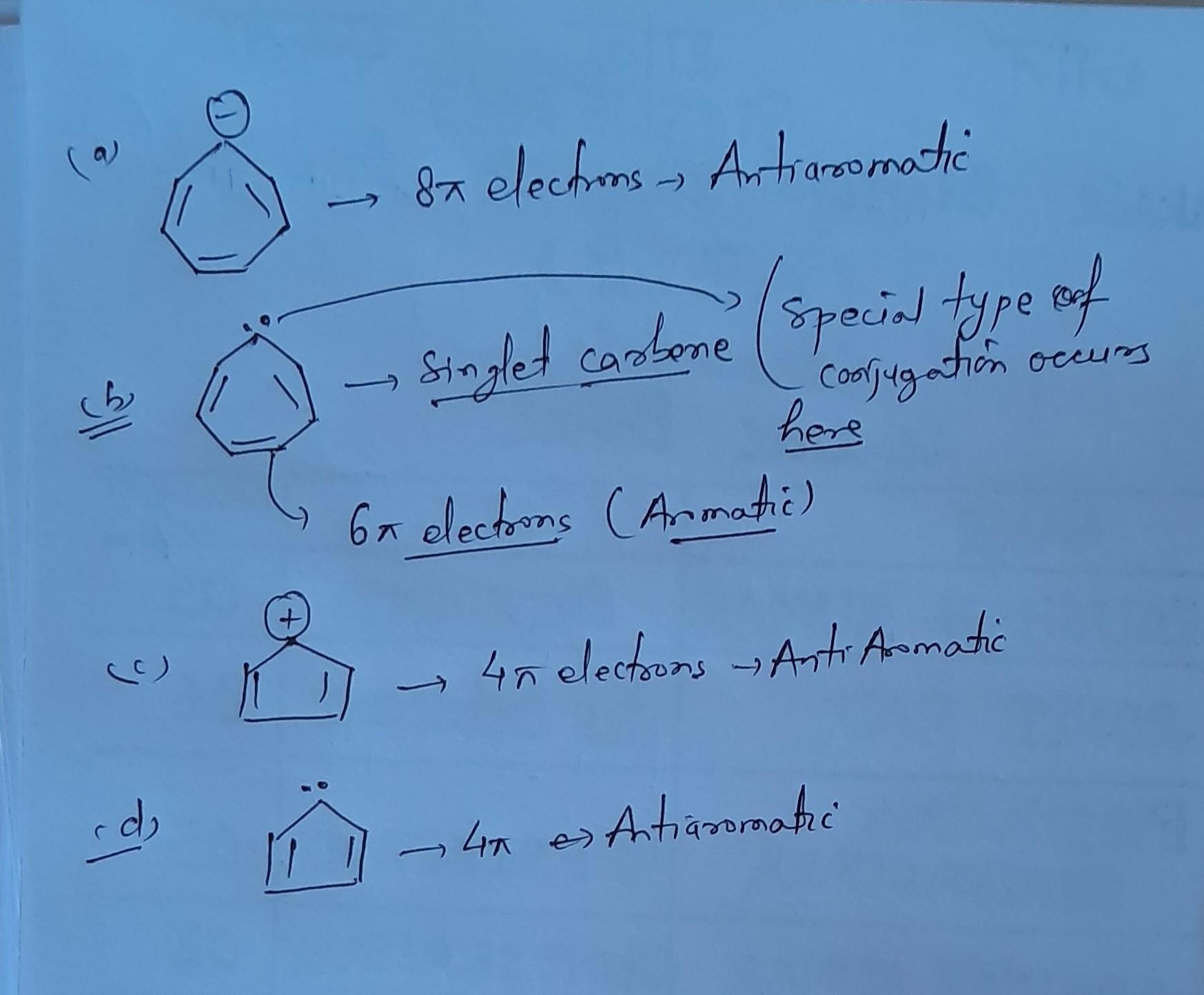
Option B is aromatic special type of conjugation occurs in case of singlet carbene....Here conjugation occurs between double bond empty p orbital double bond resonance... The electron pair resides in sp2 hybridised orbital of carbene....Not count for aromaticity....
![cropped7296862011108257352.jpg]()
![eduncle-logo-app]()
Please ask roni if any doubt
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
 >
>









Suman Kumar
option B is correct. In singlet carbene lone pair reside in sp2 hydridized orbital. so it doesn't participate in resonance