Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)
Imdadul Haque posted an Question
- IIT JAM
- Chemistry (CY)
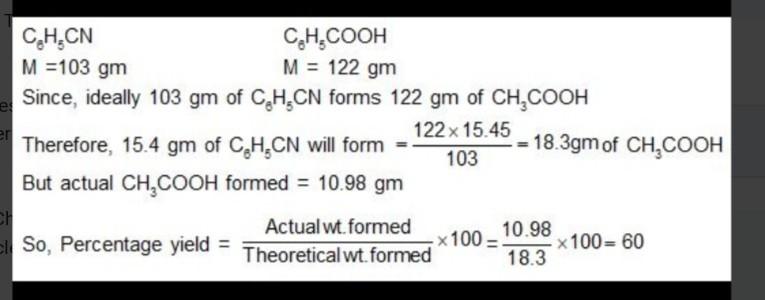
4 hydrolysis of 15.45 g of benzonitrile producted 10.98 g of benzoic acid. the percentage yield of aci1d formed is 5 acetic acid content in commercial vinegar w
4 Hydrolysis of 15.45 g of benzonitrile producted 10.98 g of benzoic acid. The percentage yield of aci1d formed is 5 Acetic acid content in commercial vinegar was analyzed by titrating against 1.5 M NaOH solution. A 20 mL vinegar sample required 18 mL of titrant to give end point. The concentration of acetic acid in the vinegar (in mol L) is according to the cquation 2N,0s(9) 4NO:() + O:(g). If the final pressure in the flask is 0.5 atm, then the partial pressure of O(g) in atm is 6. A 10 L flask containing 10.8 g of NO5 is heated to 373 K, which leads to its decomposition Given: R=0.0821 L atm K mol 7 The amount of bromine (atomic wt. = 80) required (in gram) for the estimation of 42.3 g of phenol (molecular wt, = 94g mol) is
- 0 Likes
- 2 Comments
- 0 Shares
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>








Narayanarao gundoju![best-answer]()
find the attached file