Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)- IIT JAM
- Physics (PH)
6. a and b are both classical ideal gases of diatomic molecules. the point-like atoms in a are bounded rigidly to form diatomic molecules while in b they are co
6. A and B are both classical ideal gases of diatomic molecules. The point-like atoms in A are bounded rigidly to form diatomic molecules while in B they are connected by bonds of finite stiffness. The ratio of the specific heat per molecule at constant volume of gas A to gas B is (a) 1 (c) 6/5 (b) 5/7 (d) 3/4 LISc-20101
- 0 Likes
- 6 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Ruby negi
one vibration mode active means 2 d.o.f freedom..
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Dhairya sharma
answer would be b
-
![comment-profile-img]() >
>
-
Mahak Best Answer
ans is b because in A there are 5 degre of freedom where 3 translation and 2 rotational so Cv=5/2 kt and in b there are 6 degree of freedom where 3 trans ,2 rotational and 1 vibra so E= 5/2kt +kt=7/2kt so Cv=7/2 kt so ratio is 5/7
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-











 >
>

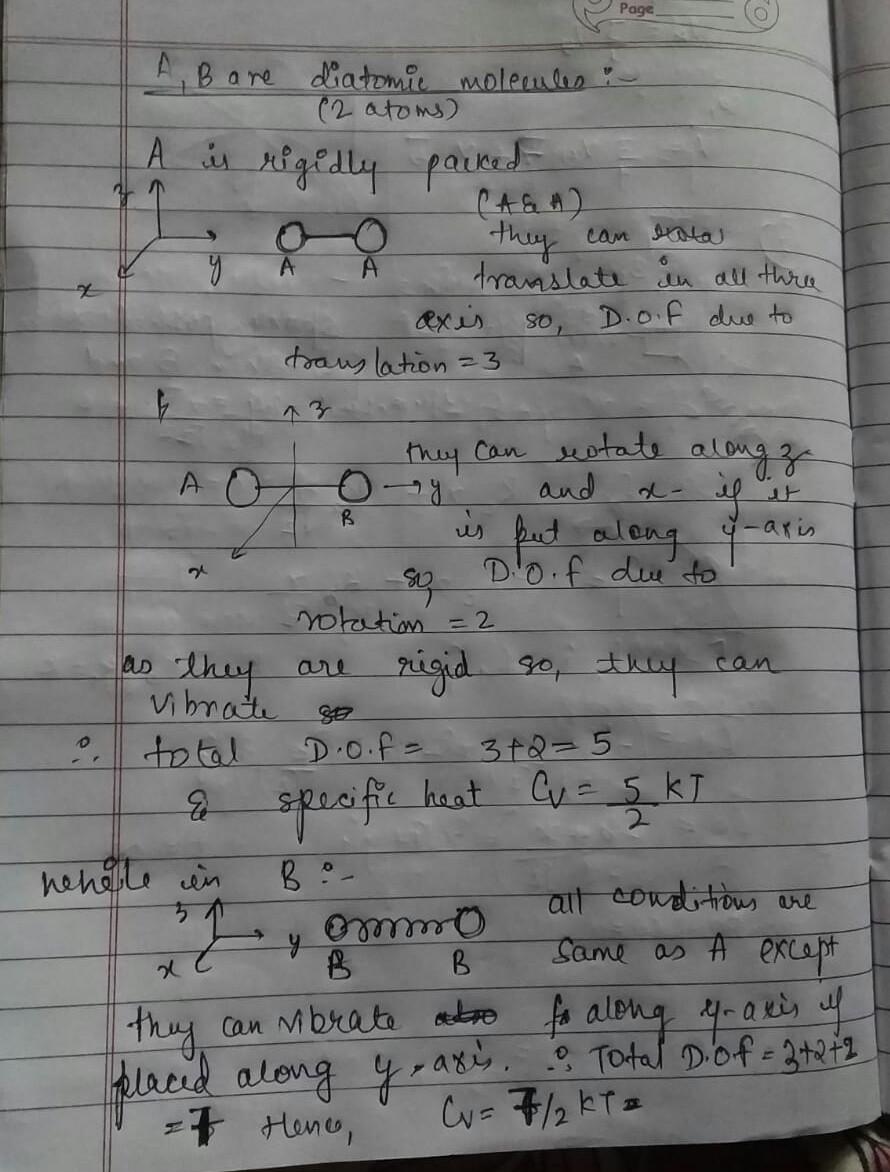
 >
>








Amrutha t
thank u