Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
Acidic order plz explain....i know in 1 and 3,1 is more acidic because lesser + inductive effect looy hodh i) cgas (ooh di) (i)
Rlooy HODH i) CGAs (ooH di) (i).ncxkhxkgxkz ljcjlxkhzgjzkhxvhkdjgzkh lhdiy
- 0 Likes
- 4 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Dinesh khalmaniya 1
![best-answer]()
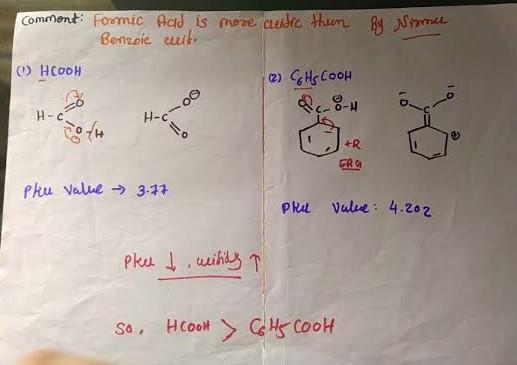
Formic acid is stronger than benzoic acid because it has a lower pKa value. Formic acid does not have such a group of electron donors and is stronger than acetic acid. The acidity is due to the stablity of congugate base. Here, In Formic acid resonance is stabilised via two equivalent resonating structure. On the other hand, in case of Benzoic acid the conjugate base is stabilised via resonance of benzene ring.
![images (3).png]()
![eduncle-logo-app]()
always resonance is more effective than inductive
![eduncle-logo-app]()
why
![eduncle-logo-app]()
kyunki overlapping polarization se Jada effective rehta right
![eduncle-logo-app]()
because resonance k during resonance energy release hoti he jo molecule ko stable krti he
![eduncle-logo-app]()
sir plz check....can I say this .
-
![comment-profile-img]() >
>
Priyanshu kumar Best Answer
formic acid is more acidic than benzoic acid.
![eduncle-logo-app]()
The acidity is due to the stablity of congugate base. Here, In Formic acid resonance is stabilised via two equivalent resonating structure. On the other hand, in case of Benzoic acid the conjugate base is stabilised via resonance of benzene ring.
![eduncle-logo-app]()
Benzoic acid is stronger than acetic acid because the electron-donating inductive effect (+I) by the alkyl group on acetic acid destabilise the conjugate base of acetic acid (i.e. the acetate ion). In contrast, benzene rings are known to be electron dense and pull electrons away from groups attached to it. order will be 1>2>3
![eduncle-logo-app]()
sir
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
 >
>



 >
>







Bhagat singh 1
answer 1>2>3