Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)
Jeremy C. Zodinliana posted an Question
- IIT JAM
- Physics (PH)
Answer any four questions from this part (each question carry 3 marks) 1. for a particular atomic state pin. what are the different possible orientation of inne
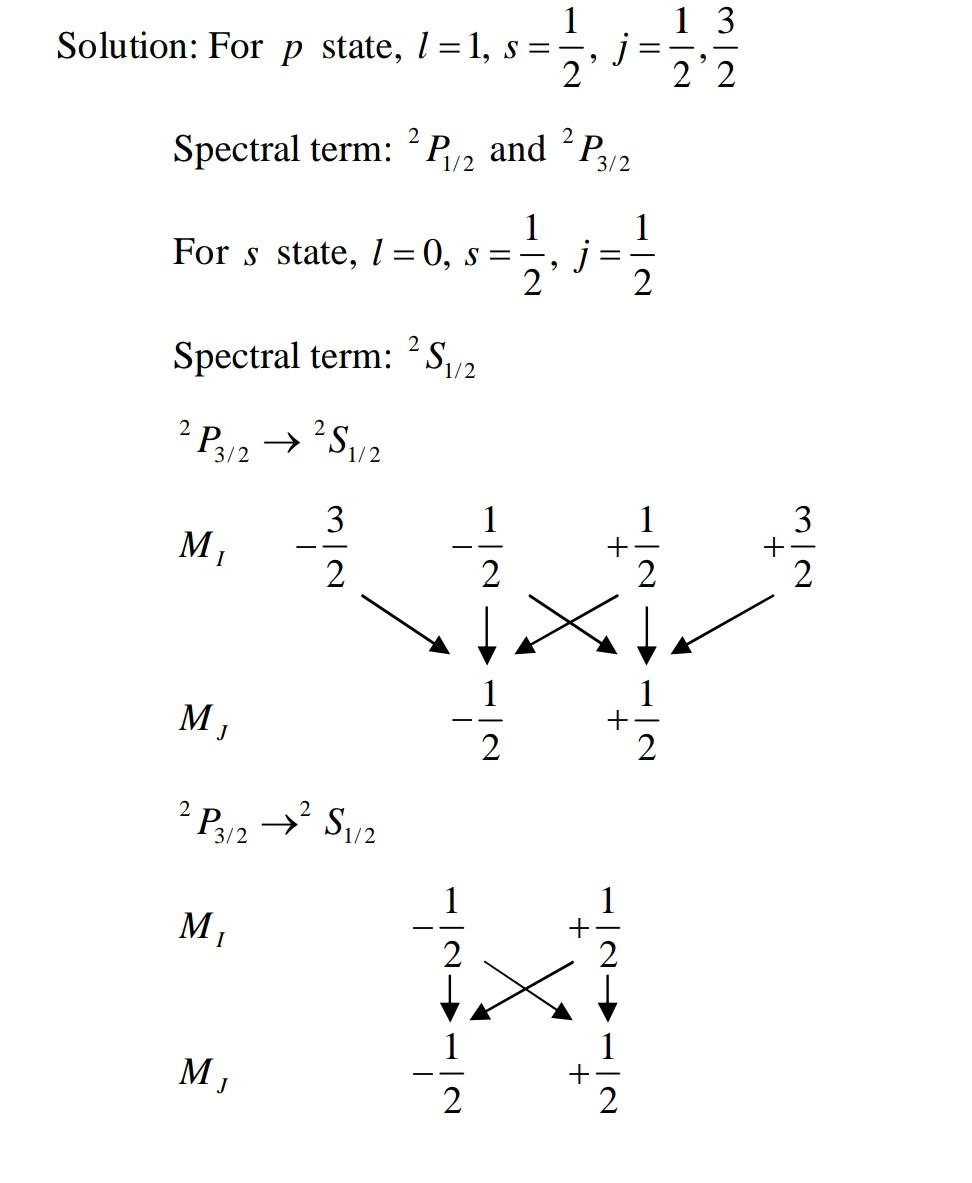
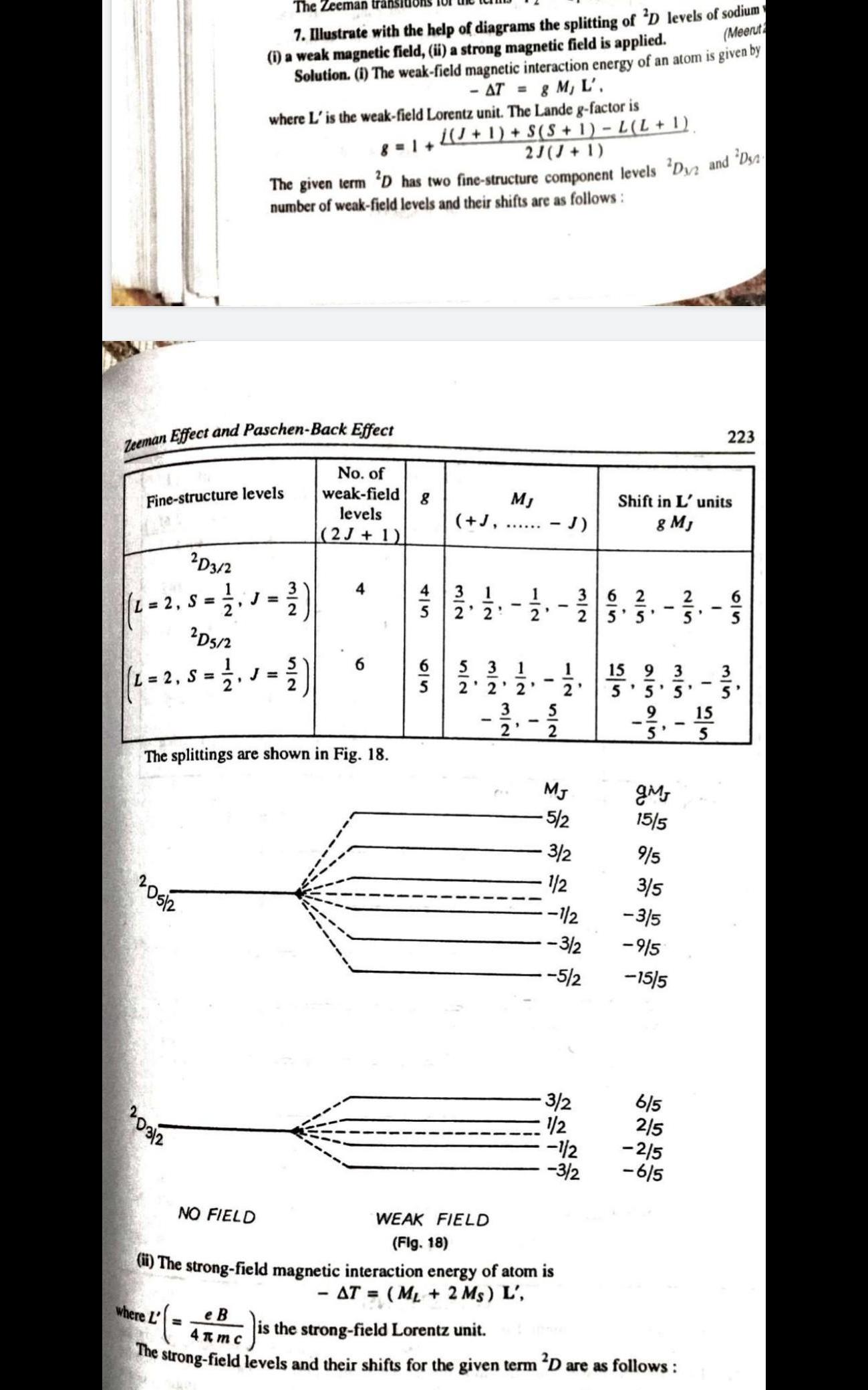
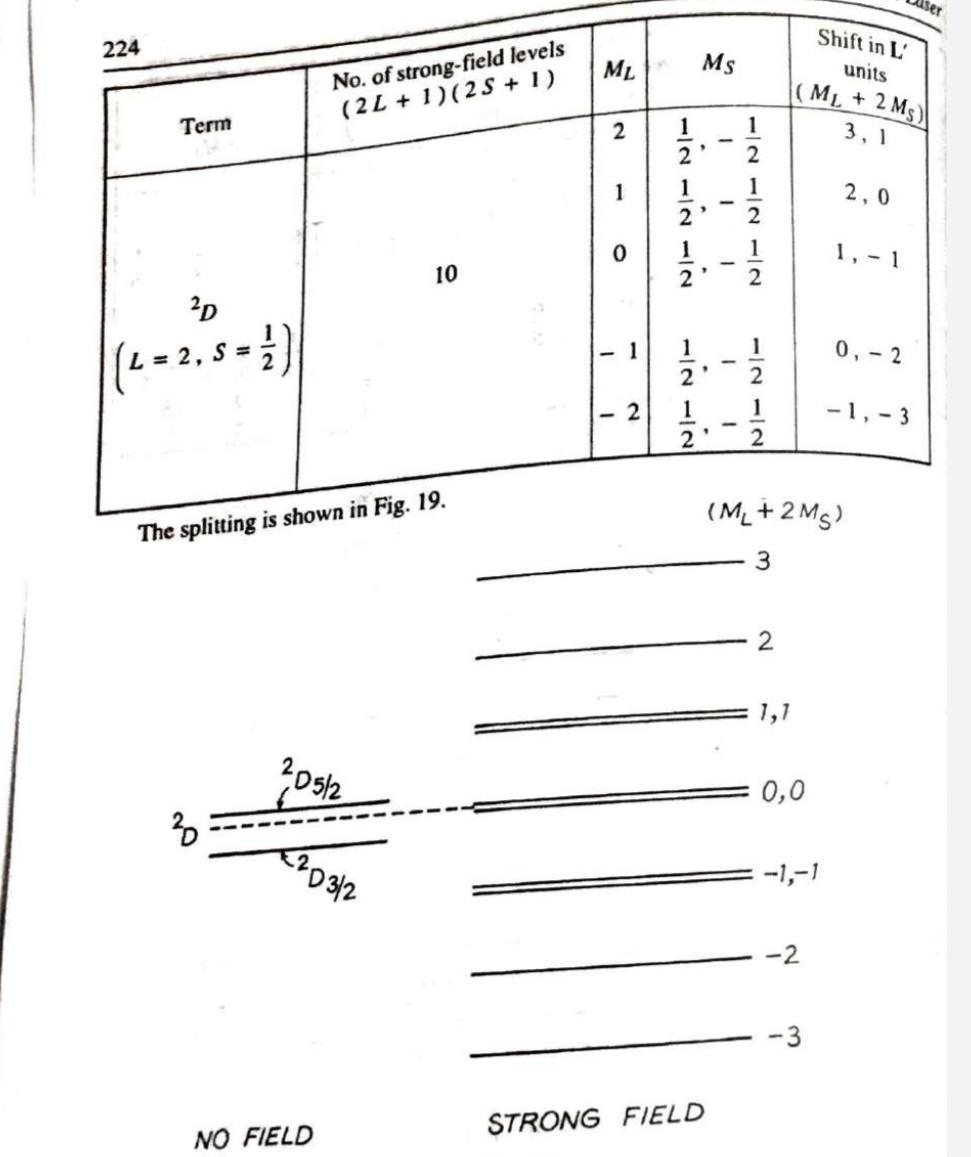
Answer any FOUR questions from this Part (Each question carry 3 Marks) 1. For a particular atomic state Pin. what are the different possible orientation of inner angular momentum in space (angle betrween inner angul ar momentum and vertical axis)? 2 Find the maximum number of electrons with all the shells fill upto principal quantum number n=3. Show that all the electrons have distinct quantumnumbers when placed in SuOng mgnetuc Tield. 3. The term of a particular atomic state is "Da What are the values of L, S, J? What is the minimum number of electrons which would give rise to this state? How did Sodium D lines split when atom is subjected to low magnetic field? Draw energy level diagram showing t and o sigma components and calculate the possible values of mg 5. A spectral line due to transition from an electronic state P to S state split into three Zeeman lines in the presence of strong magnetic field. Show with diagram that at low magnetic field the number of field/transition lines due to transition to the ground state results in ten lines. 6. 1llustrate with diagrams the splitting of D level of Sodium in weak and strong magnetic field.
- 0 Likes
- 3 Comments
- 0 Shares
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-











 >
>







Jeremy c. zodinliana
thank u so kuchhh❤️❤️❤️❤️❤️❤️