Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)
Arindam Paul posted an Question
- IIT JAM
- Biotechnology (BT)
More than one option correct
CO2 anion has which of the characteristics?
- 0 Likes
- 2 Comments
- 0 Shares
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
 >
>

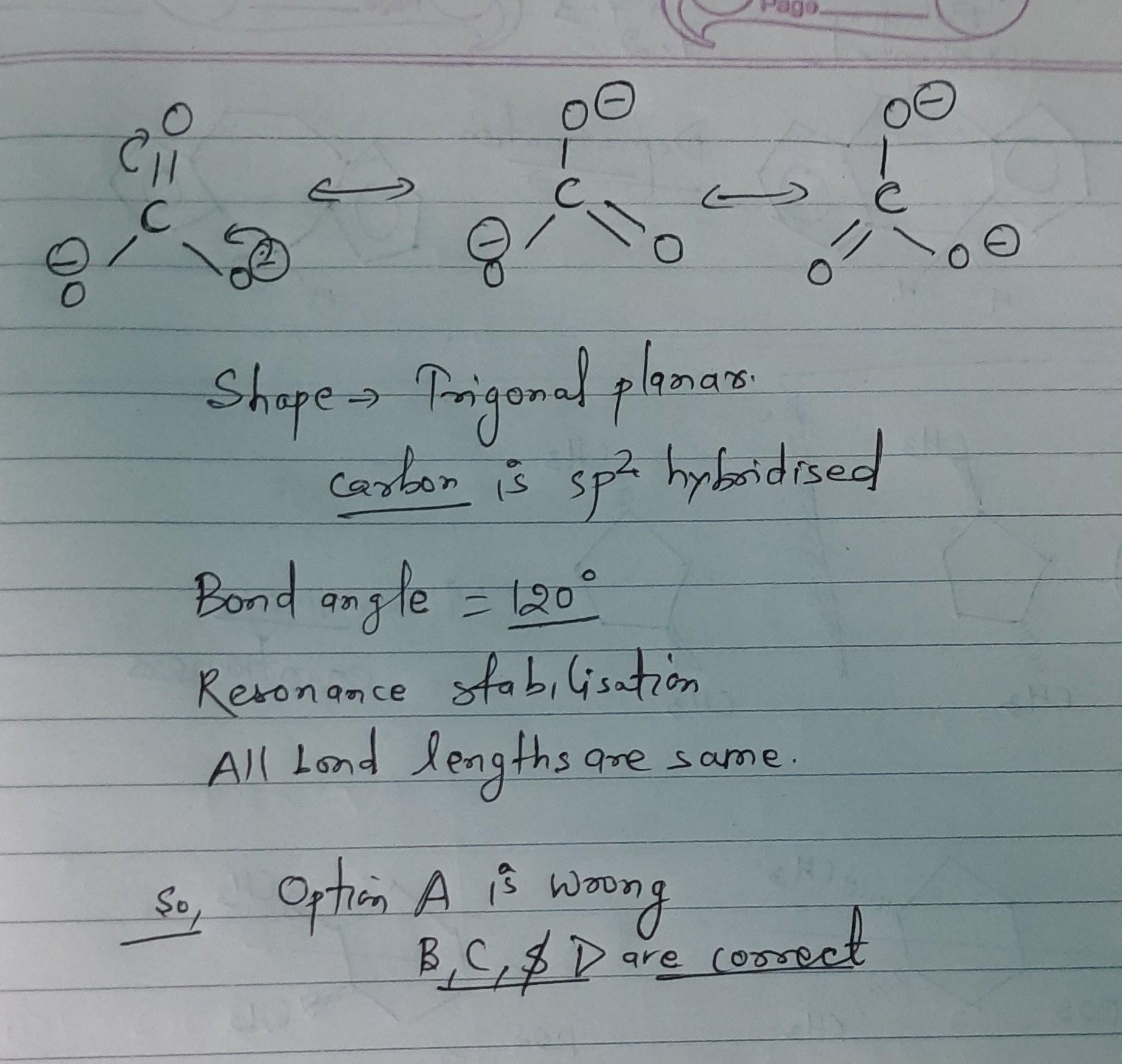








Anup Padwal![best-answer]()
All are correct. Explanations wrt Option 1) All three carbon-oxygen bond distances are about 1.28 Angstroms long. You cannot draw a Lewis structure that would suggest all three bonds are the same length. 4) Bond angle is 120 degrees as all three bonds are equal. 2) Carbon is hybridised sp2, all oxygens are equivalent. 3) resonance allows for delocalization, in which the overall energy of a molecule is lowered since its electrons occupy a greater volume, molecules that experience resonance are more stable than those that do not. These molecules are termed resonance stabilized. The stability of resonance increases with: Number of covalent bonds and Number of atoms with an octet of electrons (except hydrogen). So it shows resonance stability.