Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)- IIT JAM
- Chemistry (CY)
Give me solution...
Consider the following two parallel ireversible first order reactions at temperature T where kand k2 are the rate constants and their values are 5x 10-2 and 15 x 10-2 min, respectively, at temperature T. If the initial concentration of the reactant P" is 4 mol L", then the concentration of product R' after 10 min of reaction is to two decimal places) mol L. (Round off (Assume only P is present at the beginning of the reaction.)
- 0 Likes
- 6 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
-
Suman Kumar
Hope it will help you
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>


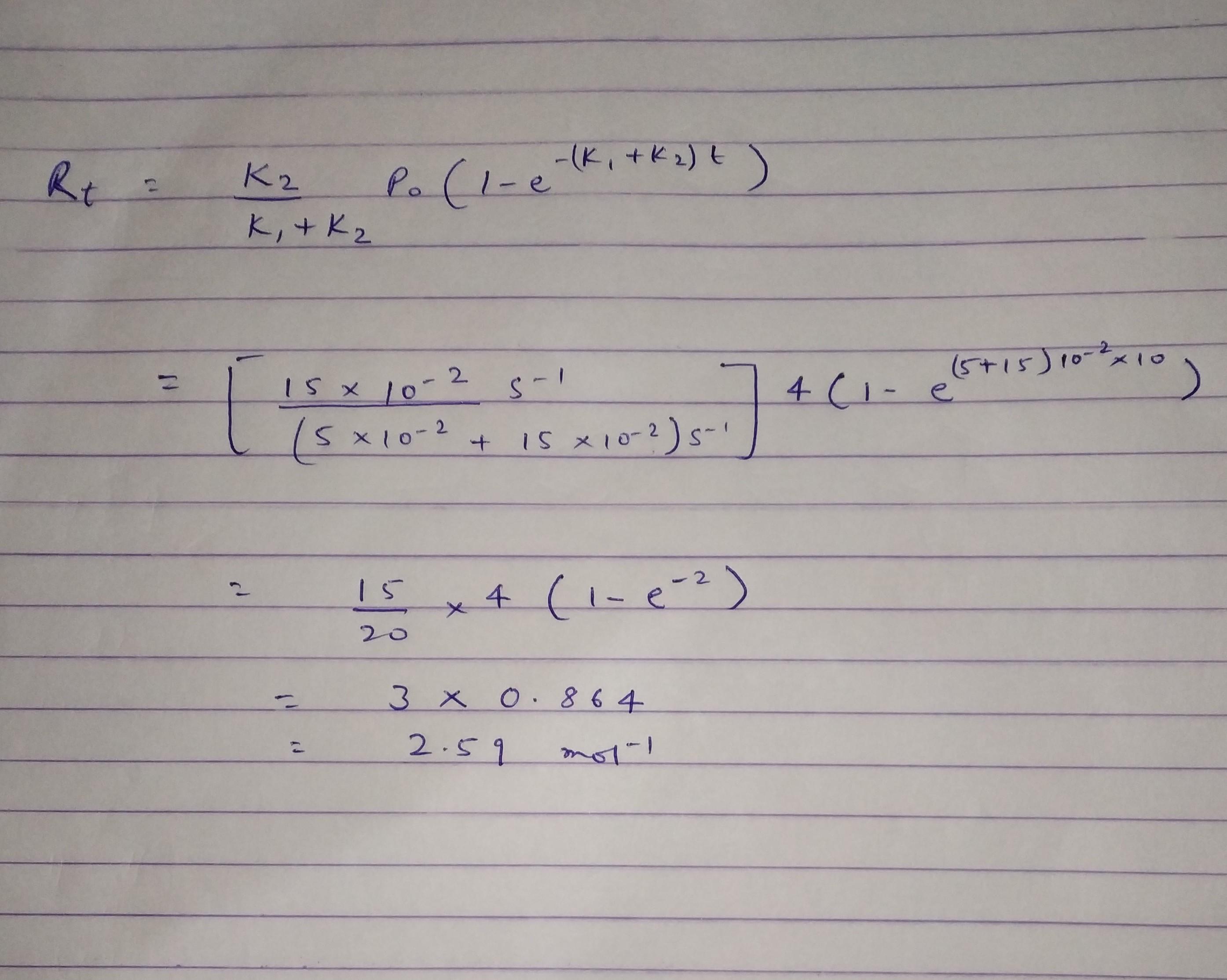








Priyanshu kumar
calculate this tejas😊
Both kumar 'S method gives same answer ...priyanshu sir thanks for explaining.. .
welcome tejas and please mark the answer too😊🙏