Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)- IIT JAM
- Physics (PH)
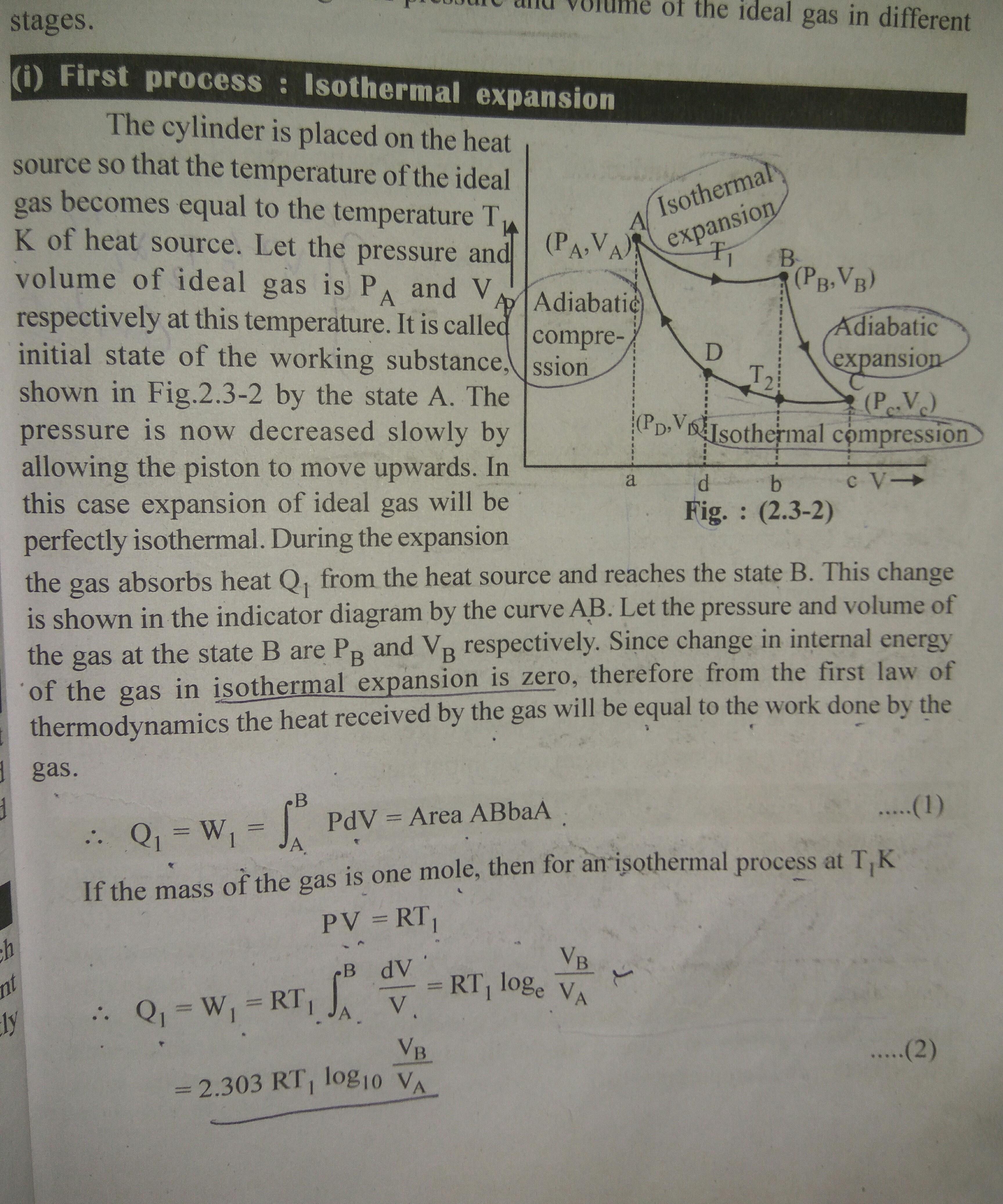
How do we calculate the efficiency of the reversible engine?
- 0 Likes
- 10 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Dhairya sharma
is it clear dear ?
-
![comment-profile-img]() >
>
Dhairya sharma
dear bcoz it's reversible process and chnz in internal energy is o so work done will equal to heat absorbed. so efficiency of ideal reversible heat engine is one.
![cropped759324628.jpg]()
![eduncle-logo-app]()
But the place where I saw this question, 1 was not in the option. The options were like 1/3, 1/6 , etc. I don't have the question right now.. I guess the engine is not reversible
![eduncle-logo-app]()
yes if it is not reversible then we get the solution.
![eduncle-logo-app]()
if we finds the solution from the above question it will give efficiency more then one which is impossible . see the attached
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Abhishek singh
you can calculate the work done by area under the curve (area of triangle is 1/2* base * height.) and the heat intake can be calculated in isobaric and isochoric process. devide the two. to get the efficiency.
![eduncle-logo-app]()
heat will be CvdT and CpdT. But we have not been given the atomicity of the gas used. so how can we calculate it?
-
![comment-profile-img]() >
>
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>



 >
>

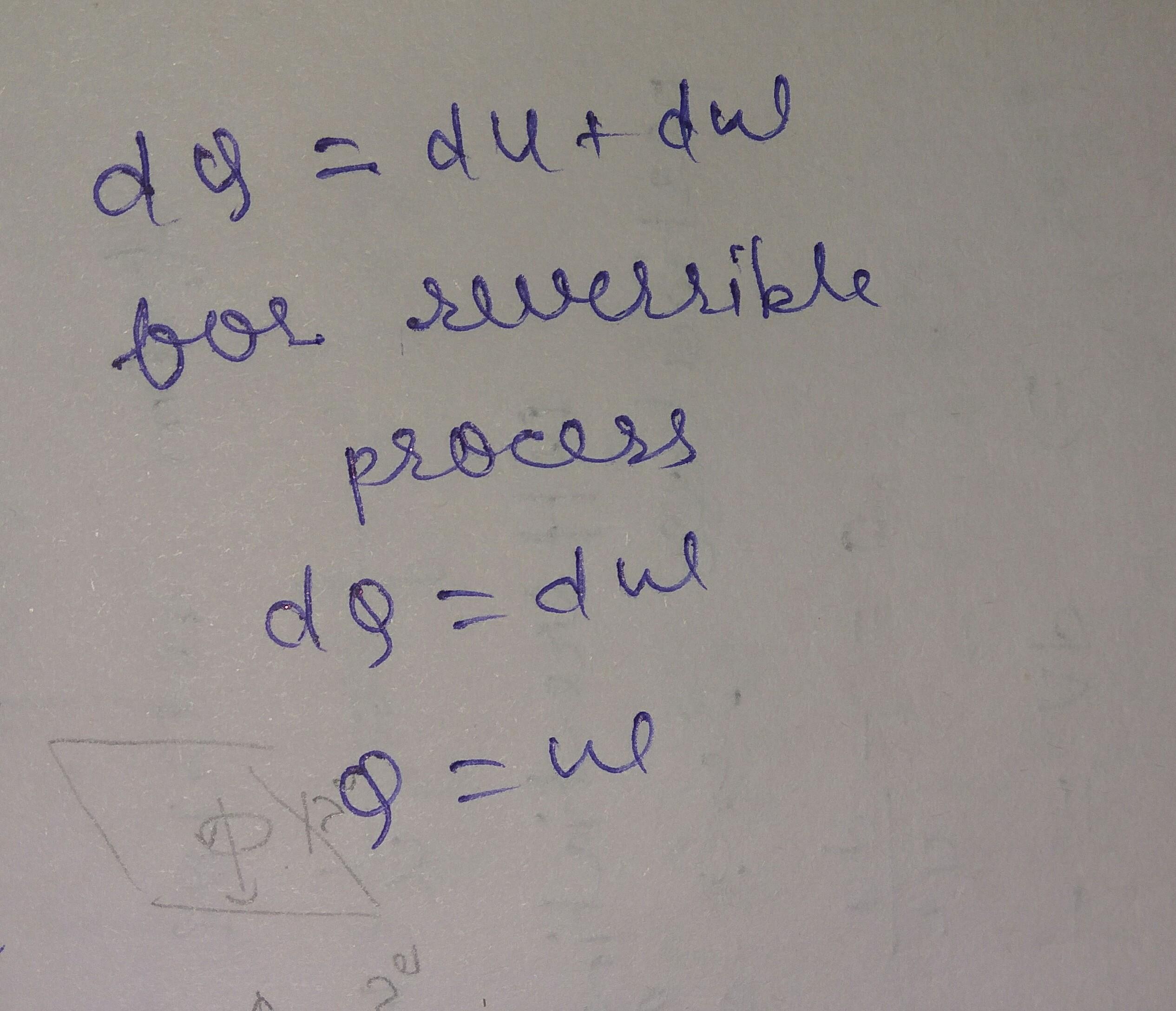




 >
>









Ruby negi![best-answer]()
A to B is isothermal process(dT=0) so temperature is same.. B to C is isobaric process for dQ=nCpdT and C to A is isochroic process so dQ=nCvdT(dW=0)...