Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
I know the answer but explanation i donot know plz explain in detail.. oh cooh cooh (16)
COOH COOH COOH (16ndbfnfmf. fnfbff fbfrbtmrvtjtktjtbjrvrnkrrjnrnrbrbrheb)
- 0 Likes
- 3 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
Achyut ranjan gogoi
![best-answer]()
3 mem ring has the highest because in 3 mem ring the %s character is max outside the ring and we know more the %s character more has the tendency to pull electron and more will be the stability of anion order will be 1>2>3
-
![comment-profile-img]() >
>
Dinesh khalmaniya 1
![best-answer]()
explanation- here we will check the groups directly attached to -COOH. EWG increase the acidity of carboxylic acid while ERG decrease the acidity of Carboxylic acid. here ring show -I effect. more the strain more is the -I. less the size of ring more is the strain and more will be the -I. hence acidiy increased with size of ring decrease
![cropped2911085557221977859.jpg]()
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>

 >
>
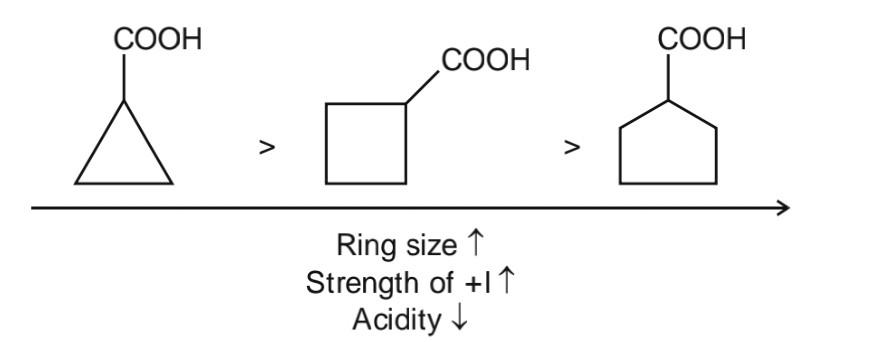







Priyanshu kumar Best Answer
Explanation:- In 3 membered cyclopropane ring there is more ring strain as compared to 4 and 5 membered ring and ring strain decreases with increase in ring size. More is the strain ,more is the % s character,and more is the electronegativity so it tends to withdraw electrons and hence show election withdrawing inductive effect(-I) and -I effect increases the acidity of carboxylic acid and with increase in ring size +I effect increases and -I effect decreases. So acidity of 1>2>3