Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)- IIT JAM
- Biotechnology (BT)
In the question like qs no 14,the r value for calculation will be given or not? there are multiple r value for different qu...(plz read description
in the question like qs no 14,the R value for calculation will be given in the exam or not? like there are multiple R value for different type of questions and I'm always in confusion...which value we should put where.. can anyone help me with this...
- 0 Likes
- 2 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Priyanshu kumar Best Answer
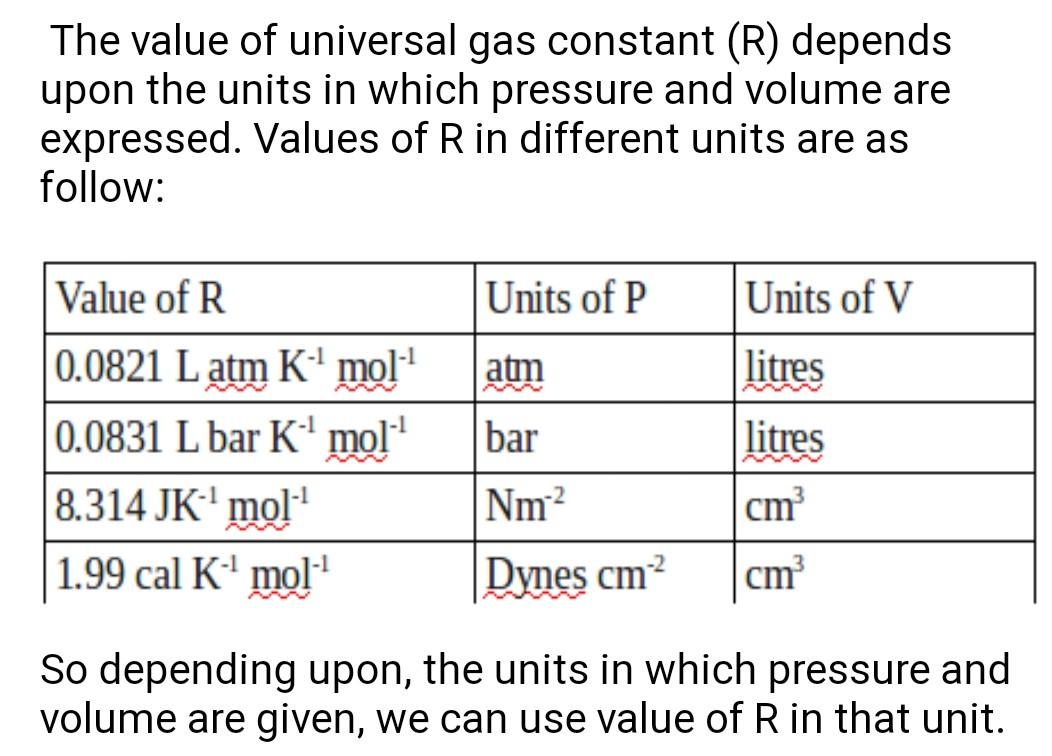
Gas constant R value depends on different pressure unit...check the pressure unit then accordingly use R value...check the below attached image for better understanding
![cropped5444764820925238619.jpg]()
![cropped2631877356496657281.jpg]()
![eduncle-logo-app]()
In ques 14 pressure unit atm is given ...so corresponding R value should be 0.0821 Latm K^-1 mol^-1
![eduncle-logo-app]()
now confusion clear na?
![eduncle-logo-app]()
yes, sir.... thanks
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-











 >
>
 >
>









Yogita Basnal![best-answer]()
Here, R = 0.08206 L atm K^-1 mol^-1. We use this value of R while solving numericals related to osmotic pressure in liquids. There is an another value of gas constant R = 8.31 JK^-1mol^-1 but value is used when we calculate Gibb's free energy. For osmotic pressure we use R = 0.08206 L atm K^-1 mol^-1 because the unit is in terms of pressure. They might give the value , they may not. Here, osmotic pressure = 0.20 x 0.082 x 293 = 4.80 atm