Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)- IIT JAM
- Chemistry (CY)
Kjtio1e aj/i1io) one mole of an ideal gas initially at 300 k and a pressure of 10 atm undergoes adiabatic expansion. () reversibly (11) irreversibly against a c
KJTIO1e AJ/I1IO) One mole of an ideal gas initially at 300 K and a pressure of 10 atm undergoes adiabatic expansion. () reversibly (11) irreversibly against a constant external pressure of 2 atm until the final pressure becomes equal to the external pressure. Find the value of ASvstem for (1) and (i) in the terms of R (heat capacity at constant volume is Cv= 1.5 R). Calculate the AH and AS for the nrocess .
- 1 Likes
- 2 Comments
- 1 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
![eduncle-logo-app]()
This is for irreversible process
![eduncle-logo-app]()
for reversible process you know this will be O
![eduncle-logo-app]()
sir ans is 0.645 to kya itni range exam me bhi di jati hai nat question me because mera bhi answers jo hai bo approx nikalte exact match nhi hite
![eduncle-logo-app]()
but method sahi hai
![eduncle-logo-app]()
ques ke according hi range milenge
![eduncle-logo-app]()
so tensn mt lo reshav
![eduncle-logo-app]()
sir entropy formula me beech me negative sign hoga
![eduncle-logo-app]()
nhi reshav sahi hai
![eduncle-logo-app]()
why reversible prcoess is zero ?
![eduncle-logo-app]()
why we take minus 0.38 ? log (300/204) = 0.38
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-











 >
>
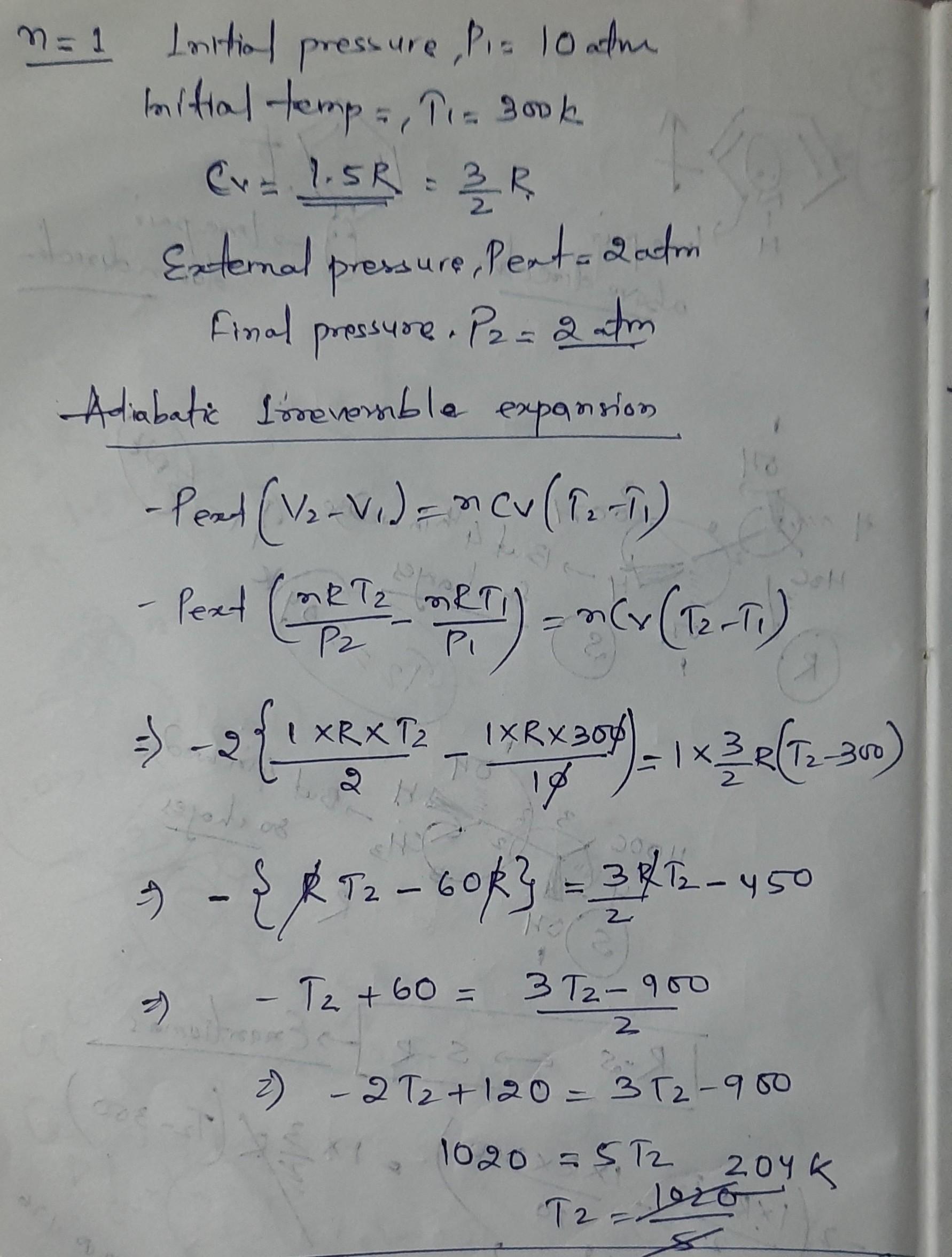
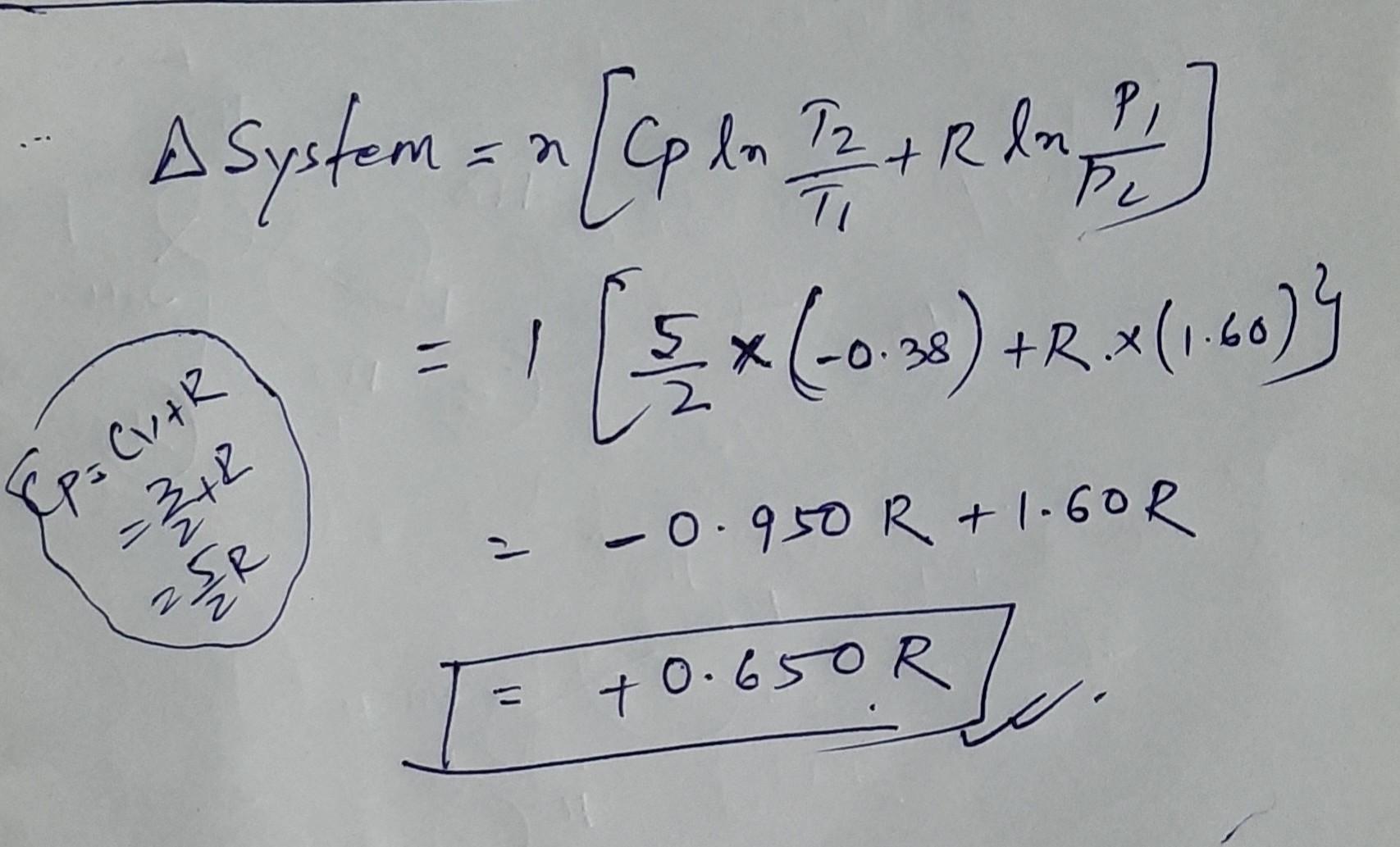








Priyanshu kumar![best-answer]()
Hope this will helps u😊