Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shib Sagar posted an Question
- IIT JAM
- Chemistry (CY)
Only solve 42 and 47
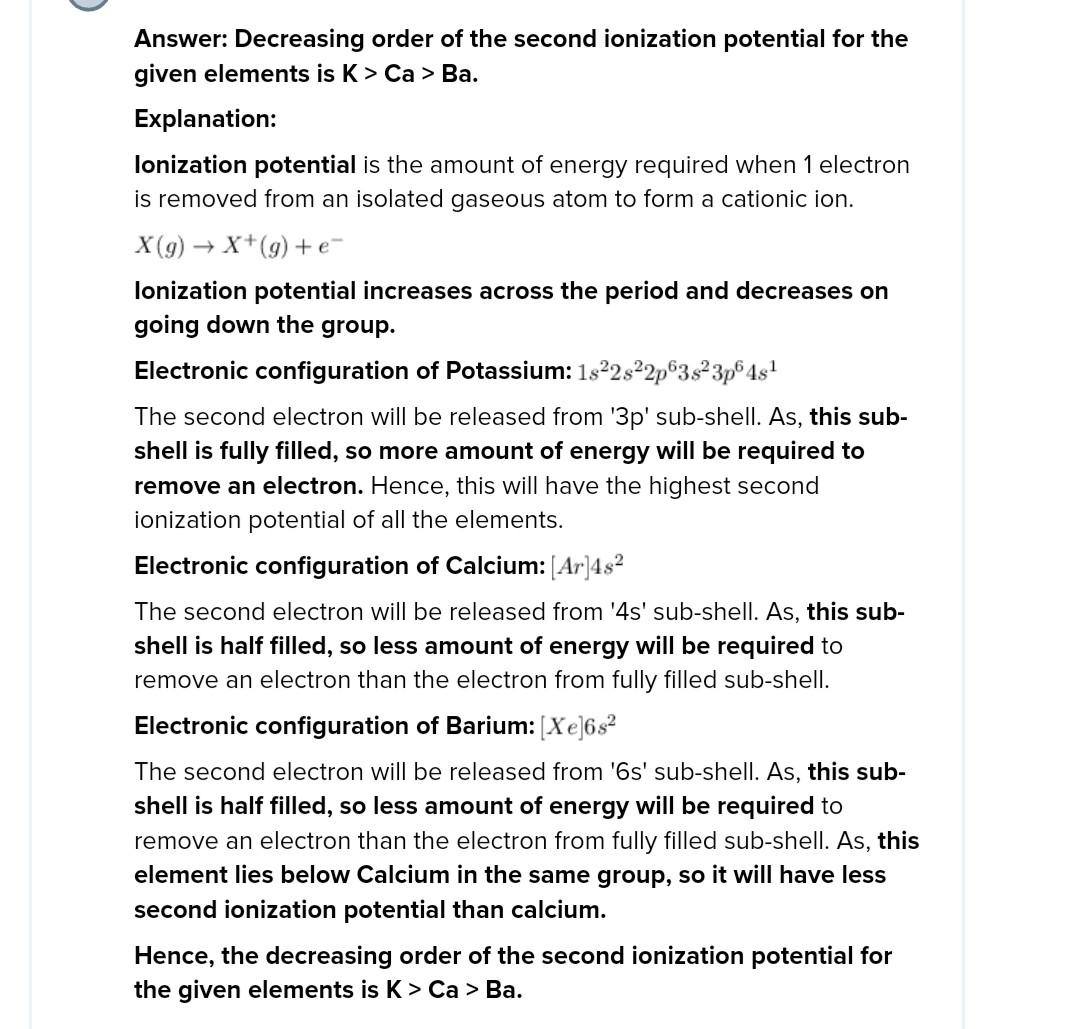
(a) Decreasing 1onization potential for K, Ca & Ba is (a) Ba> K> Ca (b) Ca > Ba> K (c) K> Ba> Ca (d) K> Ca> Ba The ionization energy will be maximum for the process. (a) Ba > Ba Alkaline earth metals always fornm dipositive ions due to (a) 1E,-IE>11 eV (C)IE, -IE,<11 eV Amongst the following, the incorrect order is (a) TE, (Al) < IE, (Mg) (c)TE, (Mg) >IE, (Na) The correct order of decreasing first ionization energy is (a) C>B>Be> Li (c)B C>Be> Li Which of the following contiguration is expected to have maximum difference in second and third ionization energies a)(1s (2sP (2p? 42/ (b) Be > Be* (c) Cs Cs* (d) Li >Li* (b) IE, - IE, = 17 eV (d) None of these 5. (b) TE, (Na) < IE,(Mg) (d) IE, (Mg)> IE, (AI) (b) C>Be>B>Li (d) Be>Li>B>C V (b) (1s (2s)* (2p)° (3s)* (3p) (d) (1) (2s (2p) () (19 (2s)P (2p)° (3s)
- 0 Likes
- 3 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Priyanshu kumar Best Answer
https://doubtnut.com/question-answer-chemistry/the-decreasing-order-of-the-second-ionisation-potential-of-k-ca-and-ba-is-at-no-k-19-ca-20-ba-56-18237888 see this video shib for 42
![eduncle-logo-app]()
sir 47??
![eduncle-logo-app]()
shib always check after removal of electrons if the atom attain noble gas configurations then there is very big jump in ionisation energies
![eduncle-logo-app]()
yes sir but in this question i m confused between a and d option
![eduncle-logo-app]()
okk let me check shib wait
![eduncle-logo-app]()
2s2 orbital is fully filled so third electron removal needs high IE
![eduncle-logo-app]()
got shib??
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>


 >
>







Dinesh khalmaniya 1
47) A because after removing one electron 2s2 configuration is obtained so it is fully filled and 3 electron is removed from fully filled orbital so it is difficult to remove so here second and third IP difference will be high
got it
sir i m confused between a and d option
why??