Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)- IIT JAM
- Chemistry (CY)
Please explain these with reason and what will be the correct answers of following?
ara8 DIts5 ng Brs and Brci 2CI- Br- ZF - Br- F>2 1- Br - Cl ABr-X bond angle (b) ZF- Br - F 13. (d) Both are isostructural Choose the correct on th (a) All C-O bonds are of equal length. bThree CI-O bonds are of equal length one longer (C) Two CI-O bonds are of same length which are longer compu (d) All are different. Which statement is incorrect about 1Cl, molecuie (a) All 1-CI bonds are equivalent (b) Molecule is polar and non-planar Correct on the Cl-O bond length in NaCIO ual length are of same length which are longer compound to other two CI-O bond length. 14. correct about 1Cl, olecule (c) All adjacent bond angles are equal (d) All hybrid orbitals of central atom having equal s-character Among the following species identify the isostruct 15.
- 0 Likes
- 7 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Dinesh khalmaniya 1
Q 13 https://doubtnut.com/question-answer-chemistry/choose-the-correct-option-on-the-cl-o-bond-length-in-naclo4-160984951
-
![comment-profile-img]() >
>
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-













 >
>
 >
>
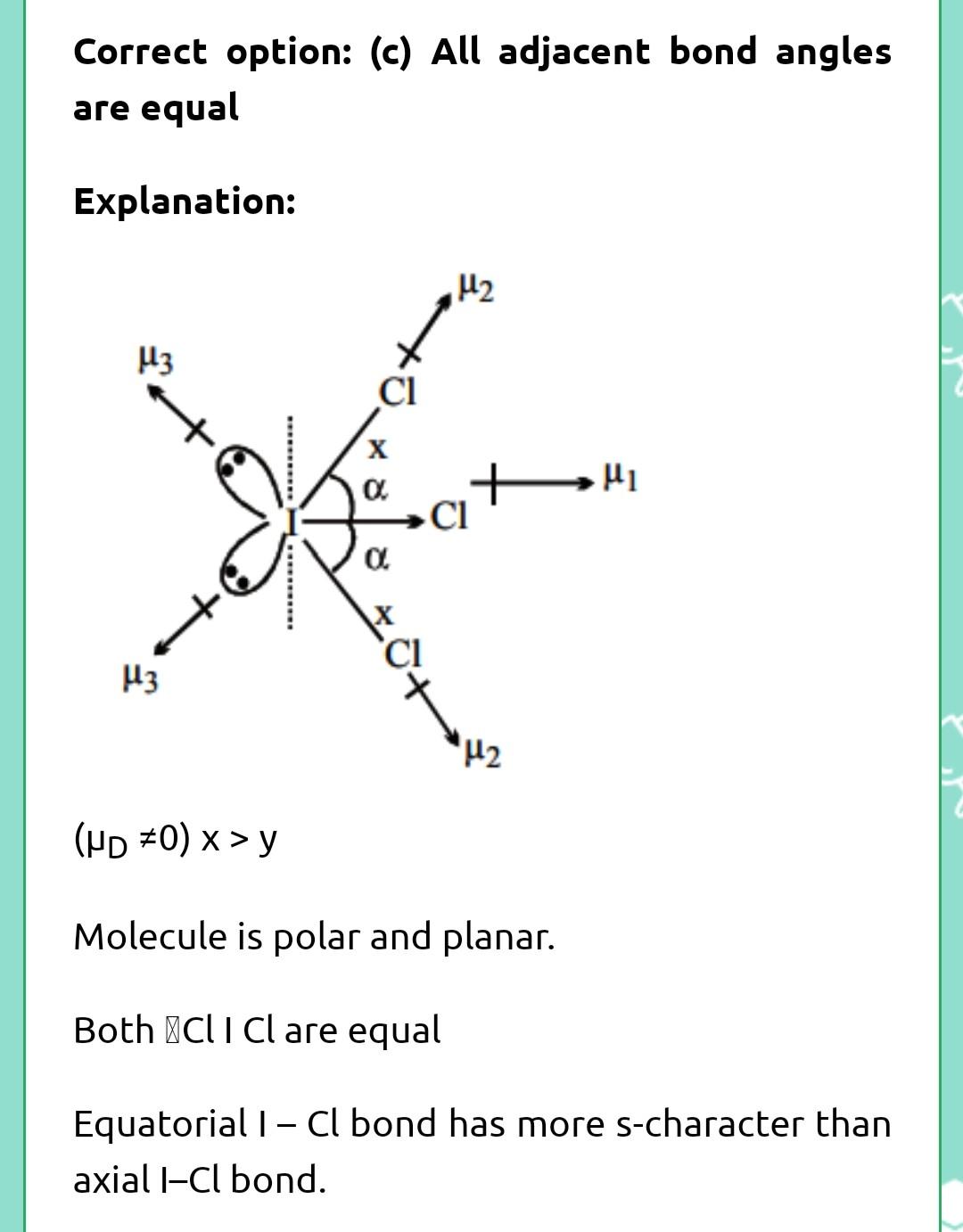
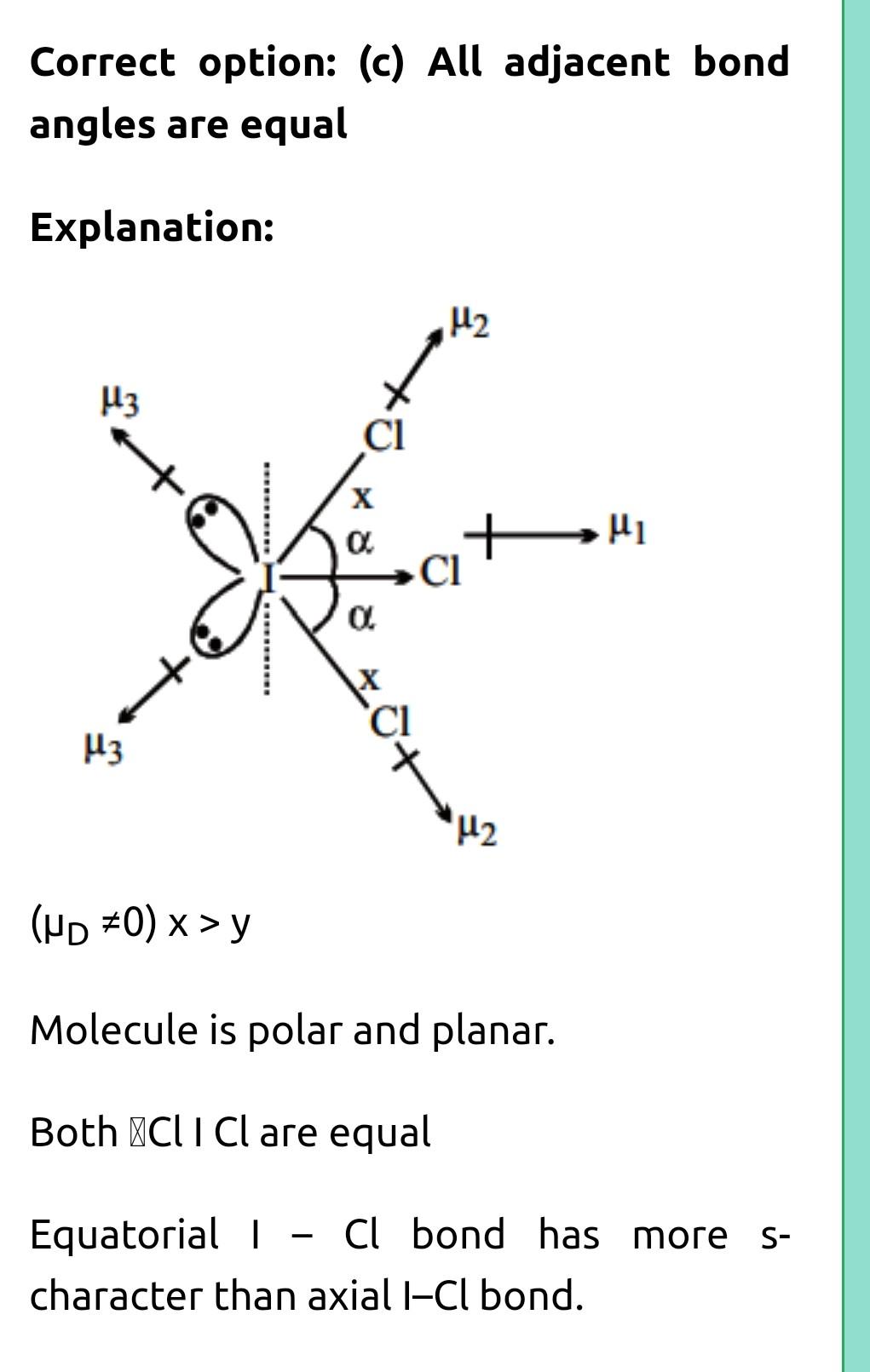







Reshav
but its answer is c
yes reshav it's C...please see my answers and mark it too if it helps🙏