Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)- IIT JAM
- Chemistry (CY)
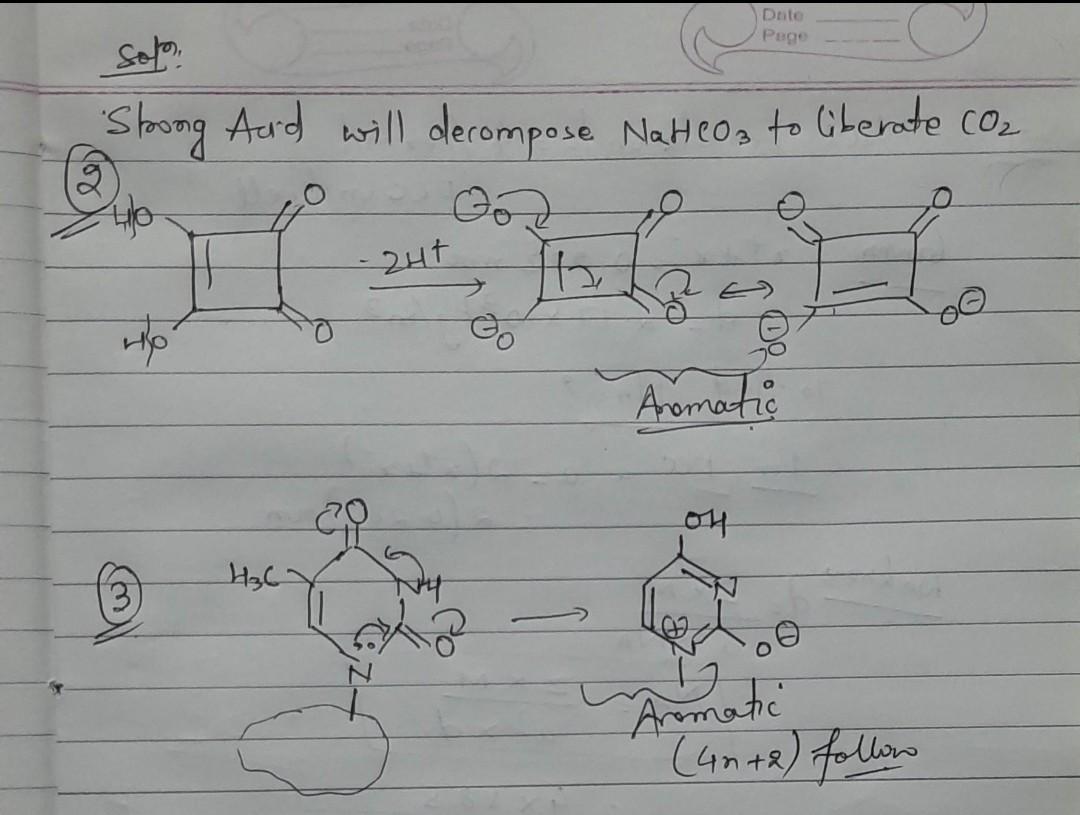
Please give solution.. and give some problems of this type...........
amoounds that reac with aqueous NaHCO, to release CO, are The compounds OH HO (A) (B) O HO OH H,C NH HO o HO N O. (C) (D) HOH,C =O HO OH HO
- 0 Likes
- 3 Comments
- 0 Shares
-
![comment-profile-img]() >
>
Dinesh khalmaniya 1
![best-answer]()
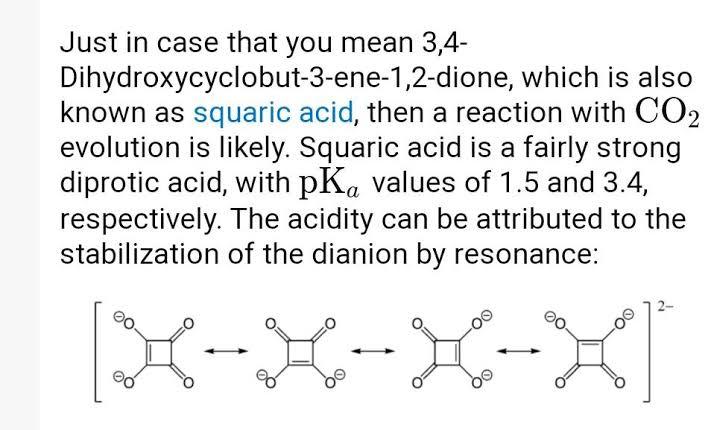
B and C. most Acidic compounds such as carboxylic acid, suphonic acid gives CO2 with NaHCO3. but some compounds such as picric acid, squaric Acid which does not give have these FG but still give CO2 because these are highly acidic. in option B it is squaric acid so most Acidic in option C it is Acidic due to Aromaticity
![images-25.jpeg]()
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>

 >
>








Amol ashok pawar![best-answer]()
Alcohols and phenols do not react with sodium bicarbonate. Carboxylic acids and enols react with sodium bicarbonate to liberate carbon dioxide. So option B and D are correct Also try to solve given problem
ans: benzoic acid, acetic acid, 2,4,6 trinitro phenol
yes perfect 👍