Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
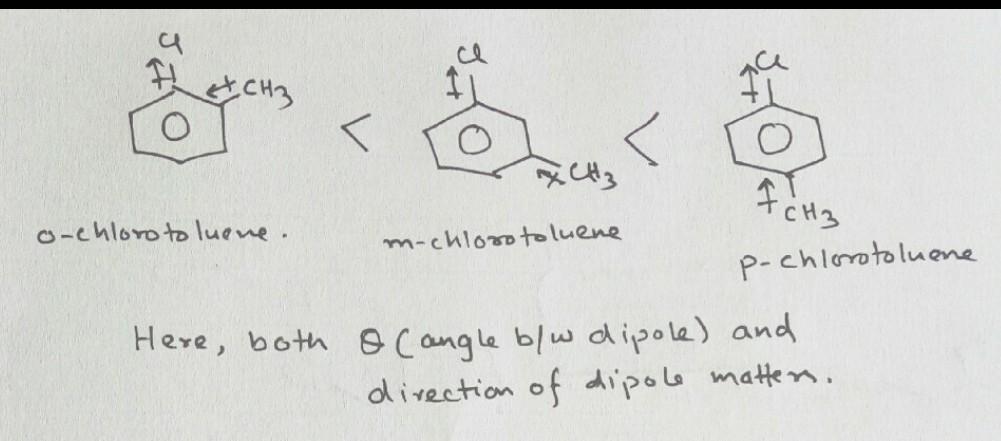
Plz explain this . t order is ci ci ch, (p) ch (b) r> q>p (c) q>r>p (d) p>r>g (a) p > q>r
e Correct dipole moment order is CI CI CH, (p) cH (B) r> q>p (C) q>r>p (D) p>r>g (A) p > q>r
- 0 Likes
- 2 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
![eduncle-logo-app]()
check bond angle between cl and ch3
![eduncle-logo-app]()
yes any doubt?
![eduncle-logo-app]()
sir
![eduncle-logo-app]()
Jaise theta bdta dipole to decrease ho achahiye
![eduncle-logo-app]()
..
![eduncle-logo-app]()
directly you can see here here the direction and angle between dipole
![eduncle-logo-app]()
here separation occurs by a larger distance in case of para
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
 >
>








Dinesh khalmaniya 1![best-answer]()
option B Explanation- Direction of dipole moment will be from higher EN to Low EN group/atom here CH3 to Cl. more the bond angle in this cass more is the dipole moment so the oder will be q>r>p
but as the theta increases dipole decreases
yes, but sweta here condition is different, yha ek hi direction me charge flow ho rha he
ap soch k dekho , agar charge rk hi direction me (180°) hoga to bilkul cancel out nhi hoga but jese hi bond angle decrease hoga kuch amount cancel hota chla jayega of jb bond angle 0° hoga to dono ek dusre k opposite ho jayege of cancil out ho jayege
any doubt?
Jo mane send kiya vo kisme use Hoga fr
jb dipole moment opposite direction me ho