Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
Plz explain this.... that a chloro substituent has a greater preference for
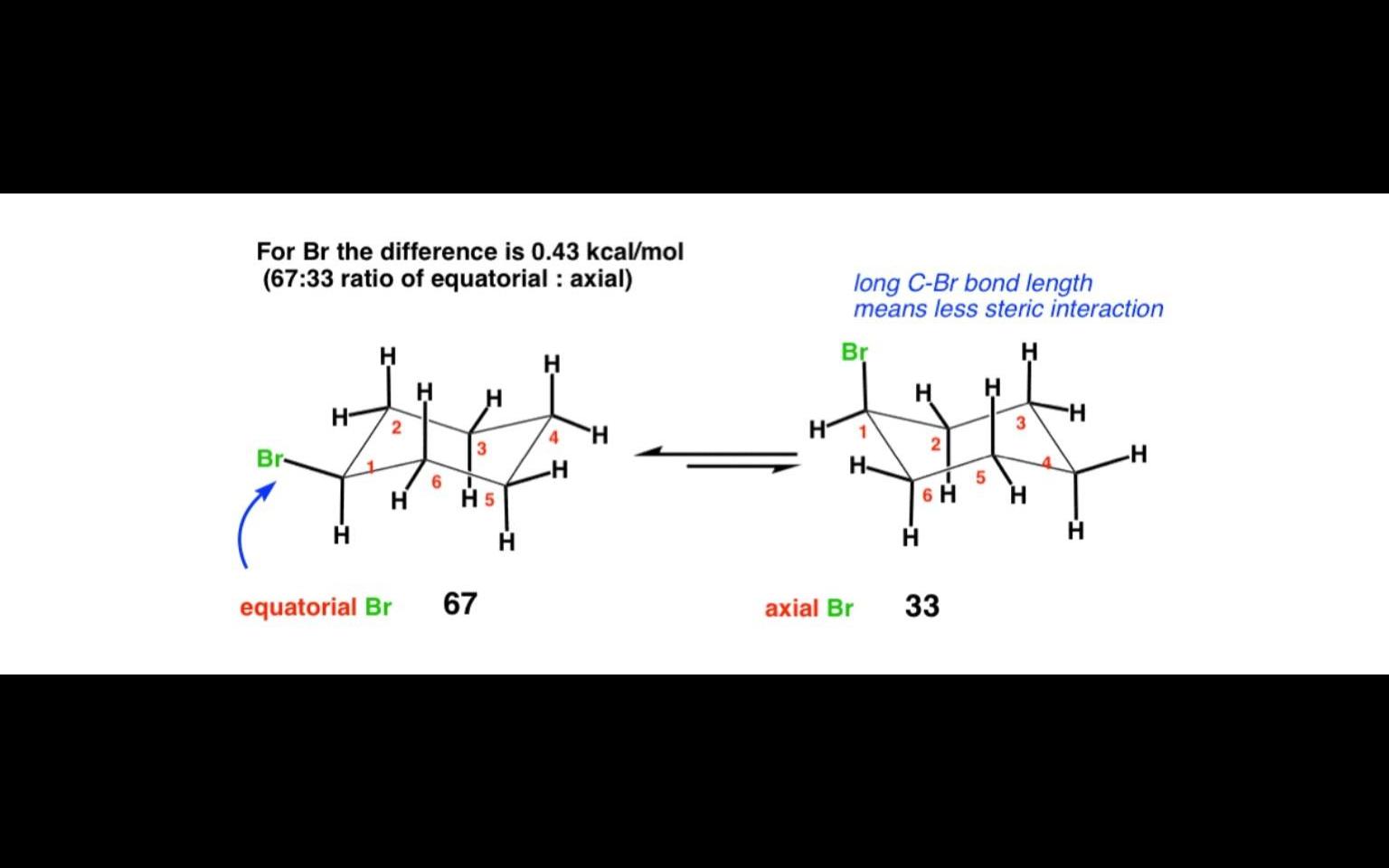
PRUDLEIVI 54 Bromine is a larger atom than chlorine, buf the eguilibrium constants in Table 2.10 indicate that a chloro substituent has a greater preference for ihe equatorial position. Suggest an ex planation for this fact.
- 0 Likes
- 3 Comments
- 0 Shares
-
![comment-profile-img]() >
>
Ashutosh singh Best Answer
Dear Shweta, “A-Values” are a numerical way of rating the bulkiness of substituents on a cyclohexane ring. The “A-Value” represents the difference in energy (in kcal/mol) between the cyclohexane conformation bearing the group in the equatorial position (more favored) and the cyclohexane conformation bearing the group in the axial position. The greater the “A-value” the higher the energetic preference for the equatorial position, and the more “bulky” the group is considered! I hope you got the whole concept of it. 😊
![cropped1383322106.jpg]()
![cropped1873897309.jpg]()
![eduncle-logo-app]()
Shweta, I'd recommend you to go through this article for 5 mins and note down some important data and points. It's very important. This - https://www.masterorganicchemistry.com/2014/07/01/substituted-cyclohexanes-a-values/
![eduncle-logo-app]()
Hope it helps.. 🙏🙏 And if it does, kindly accept the answer. 😊
![eduncle-logo-app]()
Shweta, any doubts dear?
-
![comment-profile-img]() >
>
Priyanshu kumar
![best-answer]()
Shweta the C-Br bond is longer than C-Cl bond,which causes bromine to be farther away than chlorine from the other axial substituents.... So as Br has large diameter but the longer C-Br bonds offsets the large diameter of Bromine...so chlorine has more preference than bromine for equatorial position
![eduncle-logo-app]()
got this??
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>


 >
>








Suman Kumar
Due to increase in bond length of C-Br bond Sweta