Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
Plz solve this..... q6 cooh no 2 molesa -oh nanh ho c c-h the product a will be ar of thoir o
Q6 COOH NO 2 MolesA -OH NaNH HO C C-H The Product A will be ar of thoir o
- 0 Likes
- 2 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
![eduncle-logo-app]()
got this shweta ?
![eduncle-logo-app]()
sir order ni pta chalta
![eduncle-logo-app]()
I mean kon - I dega
![eduncle-logo-app]()
kon -+ i
![eduncle-logo-app]()
Acidity check krna hai phle..coo- stabilised hai Resonating structure se most acidic
![eduncle-logo-app]()
No2 -I effect...to ye next acidic hoga
![eduncle-logo-app]()
phenoxide ion 5 resonating structure se stabilised hai
![eduncle-logo-app]()
Next alkyne
![eduncle-logo-app]()
ye SB - I effect denge
![eduncle-logo-app]()
So most acidic to least acidic order se remove honge...jitne moles given ho uske according
![eduncle-logo-app]()
carboxylate ion two resonating structures se most resonance stabilised hai
![eduncle-logo-app]()
ok
![eduncle-logo-app]()
sir
![eduncle-logo-app]()
acetylene is which group - I or + u
![eduncle-logo-app]()
i
![eduncle-logo-app]()
it show acidic character as it is sp hybridised 50% s character more electronegativity
![eduncle-logo-app]()
resonance effect dominance hote hai inductive pe
![eduncle-logo-app]()
check krna hai phle acidity ke order ko diff group ke liye....
![eduncle-logo-app]()
sir
![eduncle-logo-app]()
.
![eduncle-logo-app]()
sir
![eduncle-logo-app]()
sir
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
 >
>

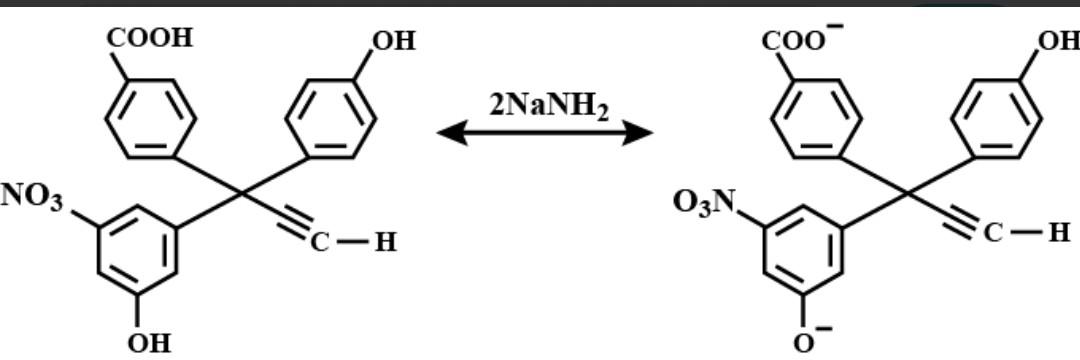








Dinesh khalmaniya 1 Best Answer
The correct order of acidity is −COOH>NO2−Ar−OH>Ar−OH>C≡CH. Thus, when only 2 equivalents of soda amide are used, first, the carboxylic group will be deprotonated, then phenolic −OH group (in the ring containing nitro group ) will be deprotonated.
no2 pehle aata I think
nhi
.....
acidity order k according hoga
NaNH2 strong base he so simply ye sbse phle more acidic H nikalega na?? or yha COOH most acidic then ArOH
ar no2 ni hai
NO2 acidic nhi he ye ArOH ki acidiy increase krta he
sir
acetylene is which group - I or + i
shweta yha pr +/- ka koi role nhi he just different functional groups de rkhe he apko acidic order yaad rkhna he
ni mujhe pta
aise for general knowledge poocha
okay
now any doubt?
Aapne btaya to hai no acetylene - I effect kyun Deta + I kyun ni
acetylene have sp hybridisation so have -I
got it shweta??
ok sir