Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
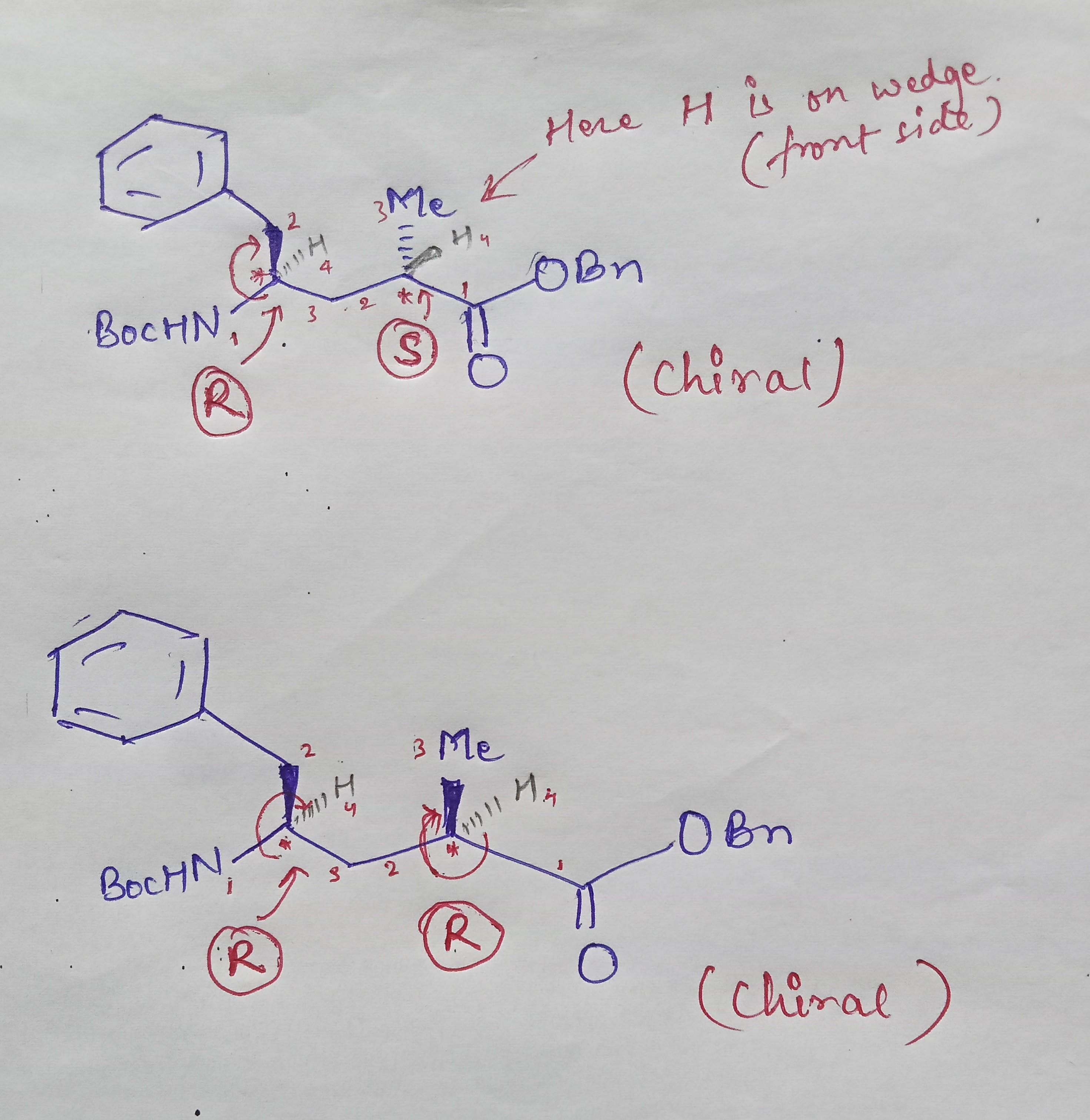
Plz tell the r and s configuration of these compound and also tell which is chiral and achiral
ed e OBn BocHN Me OBn BocHN 1b e 1a Dy LOxidation kysiyzihxohxiy
- 0 Likes
- 2 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
![eduncle-logo-app]()
sir in first two chiral carbons I think in second also r should be there if we rotate clockwise
![eduncle-logo-app]()
and sir plz tell chiral and achiral difference
![eduncle-logo-app]()
Yes shweta, you are right ..rotation is clockwise but see the position of the 4th group. It is not on dash position (below the plane of the paper). So you have to change the configuration that is from R to S. Remember while assigning priority the lowest priority should be on dash position. If not then change the configuration.
![eduncle-logo-app]()
comment if doubt remains.
![eduncle-logo-app]()
sir didn't get that why it is s
![eduncle-logo-app]()
It just a rule to assign the configuration of the optically active molecules. Also called CIP rule.
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>








Paramjeet verma![best-answer]()
If a molecule does not have any 'Plane of symmetry' or 'Centre of symmetry' then the molecule will be chiral. otherwise achiral. Chiral molecules are optically active ( rotate the plane of plane polarised light) and achiral molecules are optically inactive (no effect on plane polarised light)