Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
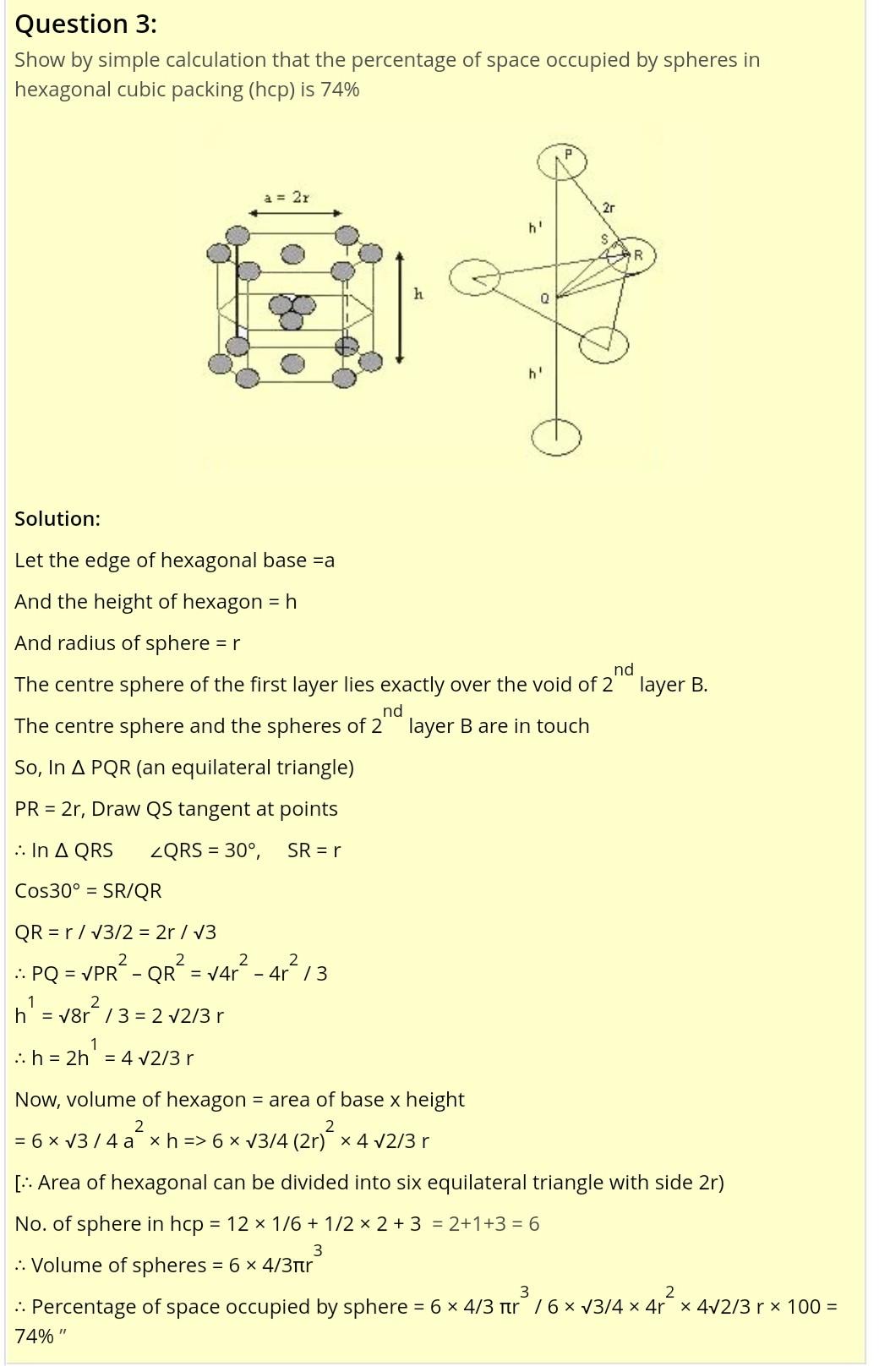
Sir can i find packing efficiency and empty space like this which i have send along with question for hcp
No. of spheres pe 21 In face-centred cubic structure (cubic close packipg). EIGURE127 FGURE 1.26 Volume of 2 spa A Fraction o- o oce 11. Sizes of As alread Dids present --- C (O Tetra Top view of face-centred cubic unit cell Face-centred cubiC unit cell The rad As sphere on the face centre is touching the spheres at the corners", evidently AC = 4r (Fig Bat from ight angled triangle ABC, 1e packin IfR is E R is AC=AB+BC=+a = a . 2a=4r or 4 a 2 olaie of the unit cell= a No. of spheres in the unit cell -s64 Sokuse of 4 spheres =4 Fraction In c hence w () (ia T sphere the ar occupied. ie, packing fractimon10Tr 13 32r/2 0-74 or % occupied 74 6 cubic packing, Hence, packing efficiency is 74%. as &packing ciiiciency of 74% Packing effcieney of hep and cep structures. Cubic close packing (cep) is same as face nitric Tha arther, cep and hep are equally efficient. He
- 0 Likes
- 3 Comments
- 0 Shares
-
![comment-profile-img]() >
>
Dinesh khalmaniya 1 Best Answer
yeh both the lattice have same packing fraction but both have different lattice so we use different methods to calculate packing fraction
-
![comment-profile-img]() >
>
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>









Shweta thakur
thanku so much for the solution