Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
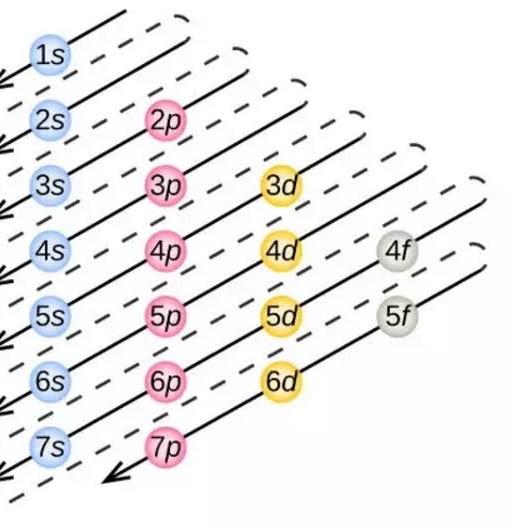
Sir plz tell that in sixty period orbital filled in according to aufbau prin. is like 6s,4f,5d ,6p but sir after filling 6s the next electron should
sir plz tell that in sixth period orbital filled in according to aufbau prin. i.e ilike (6s,4f,5d ,6p) but sir after filling 6s the next electron should enter in 4f but here next electron enter in 5d why
- 0 Likes
- 3 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Dinesh khalmaniya 1 Best Answer
election can be filled according to n+l rupe here n+l for 6s = 6, 4f = 7, 5d = 7 n+l values is lowest for 6s so electron will be filled in 6s then 4f and 5d. for 4f and 5d have same n+l values. so we will check the value of n.. election will be fill first in that orbital which has lowest value of n. so the electron filling order will be 6s , 4f and 5d
![cropped9165359909237081911.jpg]()
![cropped43005456697214426.jpg]()
![eduncle-logo-app]()
but sir in 6th period after filling 6s filling of 5d starts this am asking how it happen
![eduncle-logo-app]()
but according to rule after 6s electron will fill in 4f not in 5d. check the triangle
![eduncle-logo-app]()
sir
![eduncle-logo-app]()
I am sending u pic plz check
![eduncle-logo-app]()
electrons are filled according to Afbau principal. and according to thiselectrons are filled into atomic orbitals in the increasing order of orbital energy level. According to the Aufbau principle, the available atomic orbitals with the lowest energy levels are occupied before those with higher energy levels. and energy can be calculated by using n+l rule
![eduncle-logo-app]()
but sir 6 th period main after barium they filled elecyron in 5d not 4f
![eduncle-logo-app]()
do you have any reference regarding this because i didn't get thia info anywhere
![eduncle-logo-app]()
sir I have sentt the pic
-
![comment-profile-img]() >
>
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>


 >
>











Dinesh khalmaniya 1![best-answer]()
check the explanation