Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shib Sagar posted an Question
- IIT JAM
- Chemistry (CY)
Solve the 1st question and here i marked the other questions answer those r not matching with the answer sheet pz clear my doubt
Aolp otoms of x cg) me convernted into ) Pole tiol Cmd eleetrr o
- 0 Likes
- 3 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
 >
>

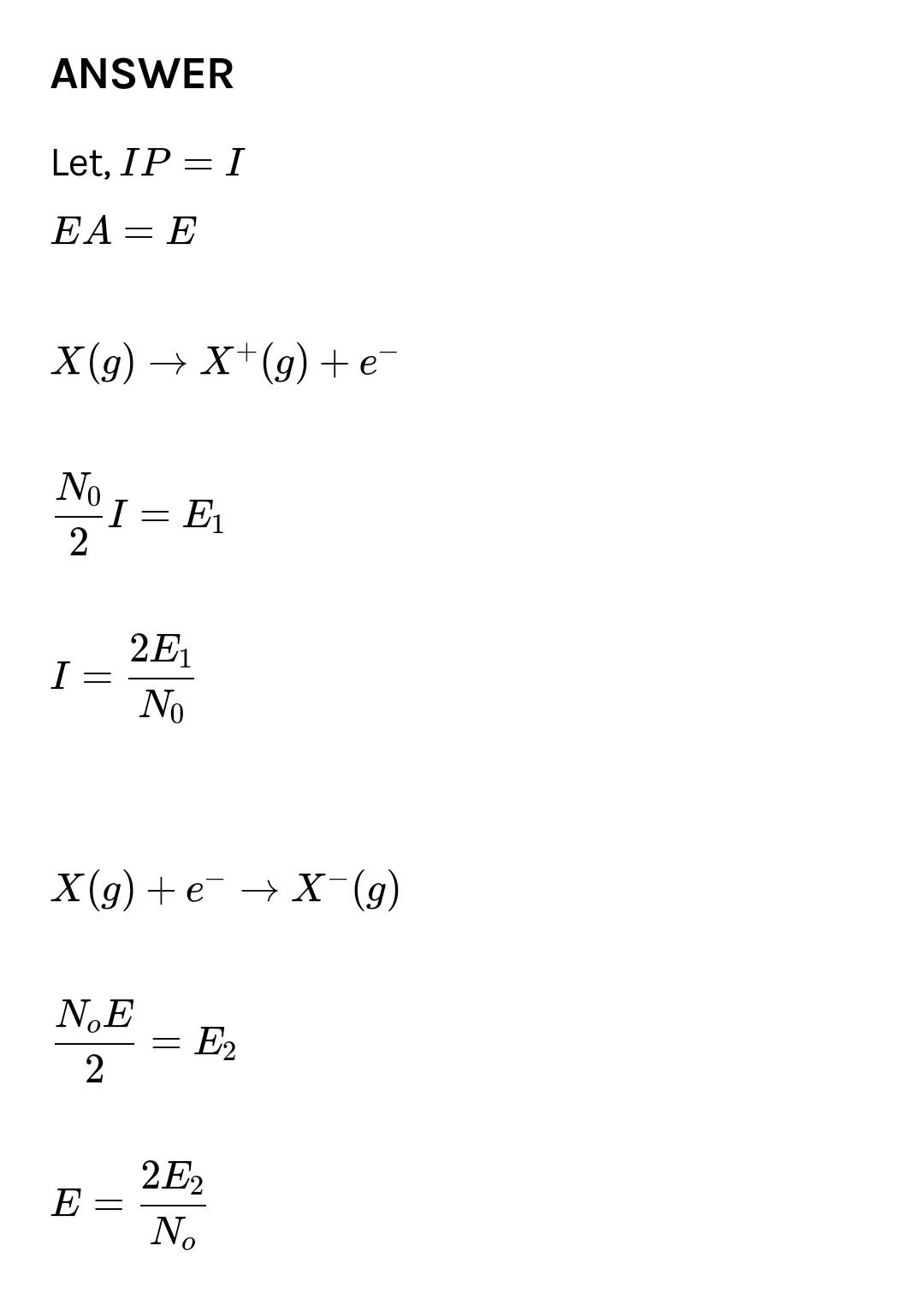









Priyanshu kumar
shib for ques 10 the correct option is option C..as the chlorine has more tendency to accept electrons...so its electron affinity value is more negative,more energy release, the first option is the case of ionisation enthalpy which is endothermic(energy needed),the 2nd option is the case of 2nd electron gain enthalpy of oxygen which is positive and the 4th option is the case of electron affinity but its value is less negative than value of chlorine..So correct option is option C
for ques no 14...as you know shib ionisation enthalpy whether first second,third etc is always positive means endothermic, ...the case arises for 2nd electron gain enthalpy which is always positive that is endothermic...the first electron gain enthalpy is always exothermic but for noble gases it has zero or positive value...so considering this you can say first electron gain enthalpy has tendency to show both endothermic and endothermic....
for ques 18...as you know second IP is the energy needed to remove a second electron from each ion in 1 mole of gaseous 1+ ions to give gaseous 2+ ions.so check after removal of one electron which compound has more stable structure...those which have more stable structure then they have more second IP..your configuration is not clear...so check on this concept...it helps
hope you got it😊🙏
please accept the answer if it helps😊🙏
shib got it??
dear shib please respond