Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)
Jeremy C. Zodinliana posted an Question
- IIT JAM
- Physics (PH)
urgent
th H-atom. Due to this collision, H-atom gets ionized. What is the maximum kinetic energy of emitted electron? 3. For an electron revolving around the nucleus in a clockwise direction, in the presence of magnetic field in the z-axis direction, what is the action of Torque on the angular momentum of that electron? Explain with diagram. 4. The energy of excited hydrogen atom is -3.4 eV. Calculate the angular momentum of the electron according to Bohr theory (h = 6.63x10** Js) 5. The wavelength of the first member of the Balmer series in the hydrogen spectrum is 6563 A. Calculate the wavelength o the first member of Lyman series. 6. For n =4, find the possible values of angular/azimuthal quantum number and draw the orbit diagram indicating their quantum numbers.
- 0 Likes
- 2 Comments
- 0 Shares
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
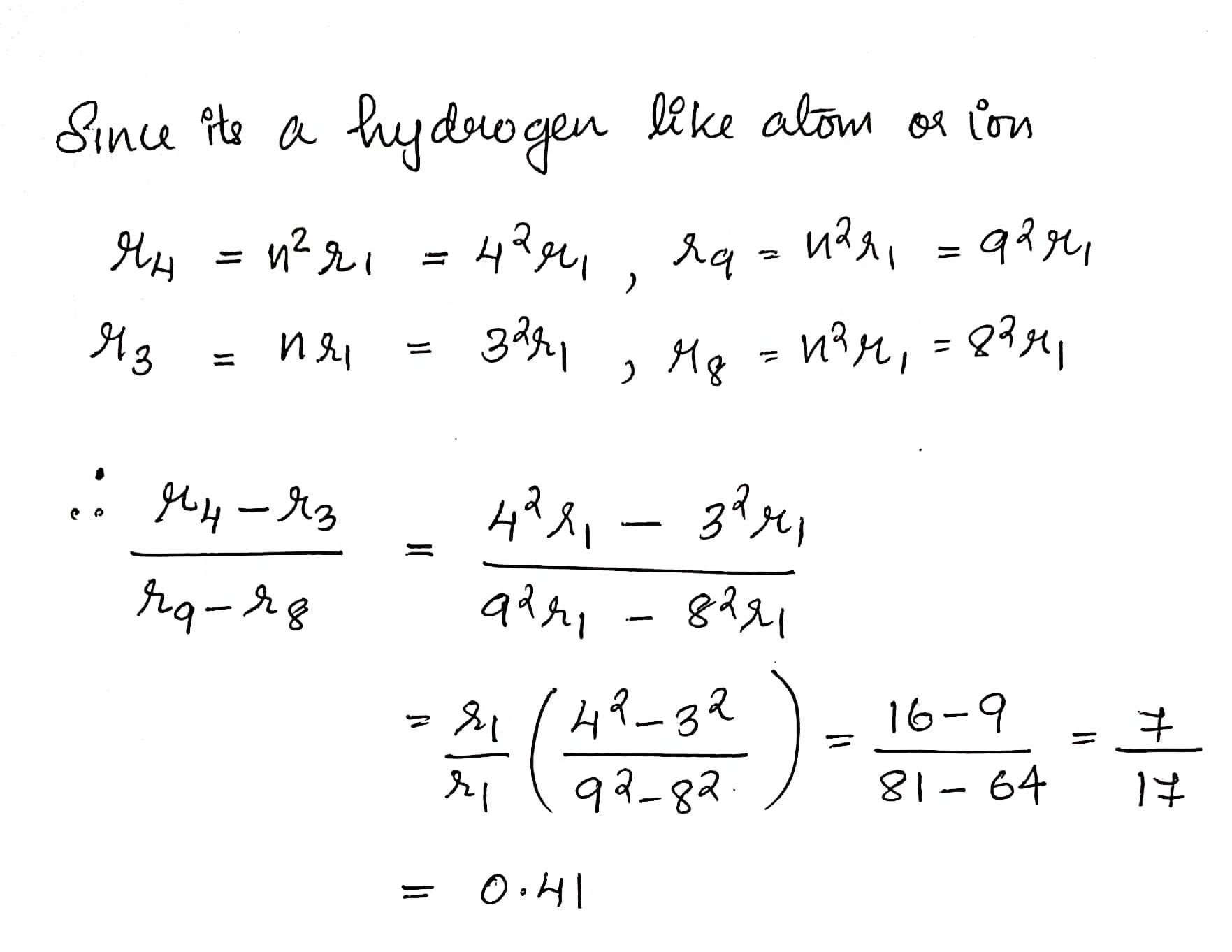









Jeremy c. zodinliana
thank u so much ...youre the goat💗💗💗💯💯💯💯