Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)- IIT JAM
- Physics (PH)
Thermodynamics
kindly provide answer with each one explanation?? .
- 0 Likes
- 11 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
Nikihi
yes sir why D not C
-
Himanshu Pandey
shanu sir i am also thinking like you only the whole problem is between option C and D they are accepting D not C ???why
-
Himanshu Pandey
Somnath sir ok but why option c should not be the answer it is the definition of entropy which is also state function
-
Himanshu Pandey
no mam answer is A,B& D i want to know how with explanation too
-
![comment-profile-img]() >
>
Ruby negi
this is the explanation. hope this will explain u how dU=0 for cyclic process.. similarly for entropy . Note entropy and internal energy are point functions , does not depend on path followed by process that's why entropy and internal energy is zero for cyclic process... as dS=dQ/T so option a,b,c correct..
![cropped-668325822.jpg]()
-
![comment-profile-img]() >
>
Somnath
Since Internal energy and entropy both are state function so change in intetnal energy and entropy is zero.Also integration of dQ/T less than equal to zero is the Clausius inequality (see any book for clarification). So there are three correct options a,b, and d.
-
![comment-profile-img]() >
>
Shanu arora
OPTION A,B and C are correct because A: U is astate function only depend on initial and final position(final - initial) in integral not on path (like dQ or dW) ,so the cyclic integral is zero ( final - final=0) . B : S is also state function , C: this is defination of dS ( entropy ) then again S is state function .
-
![comment-profile-img]() >
>










 >
>

 >
>

 >
>
 >
>
 >
>
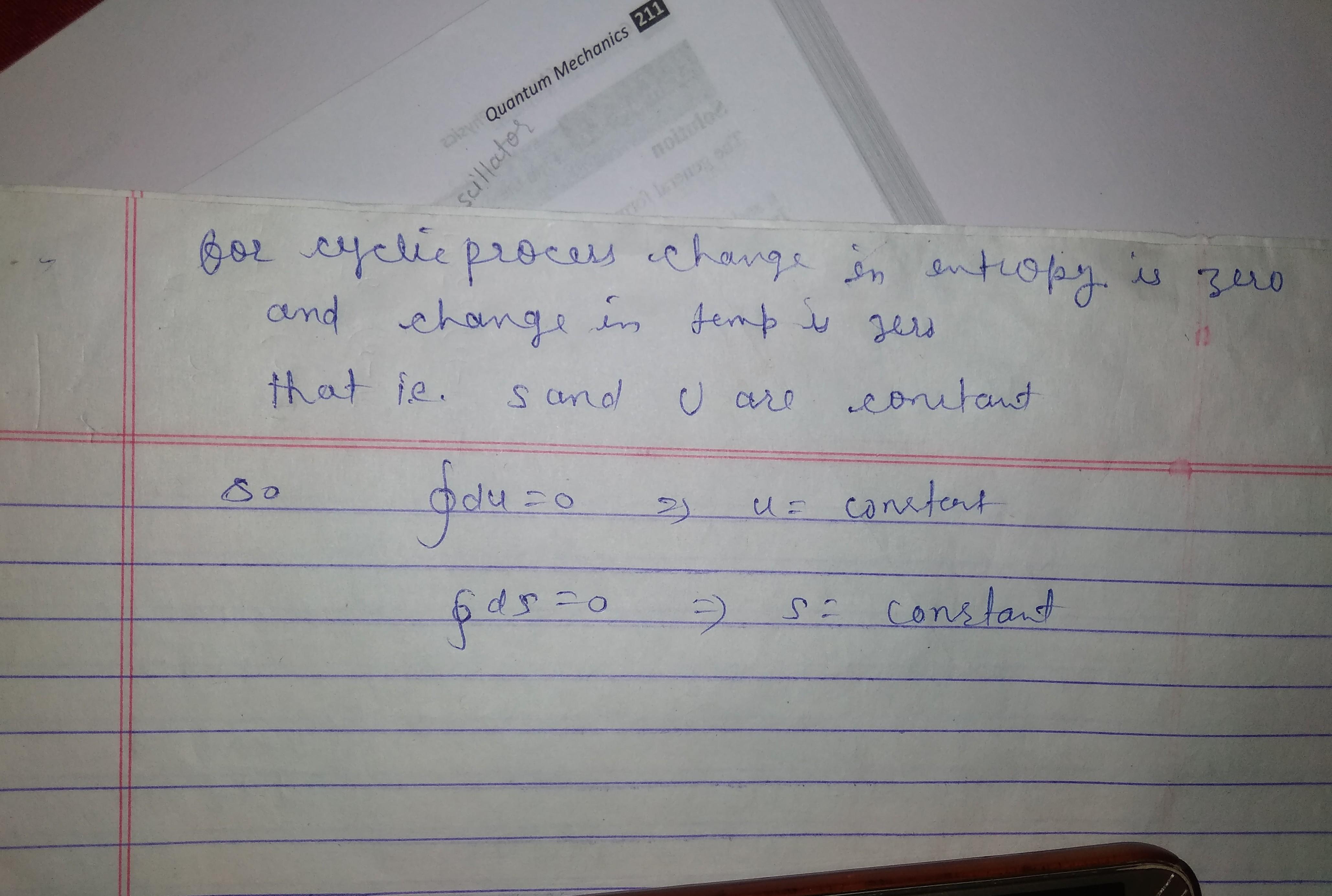








Abhishek singh Best Answer
As far as I understood your doubt. you are getting confused among option C and D, right? if yes, the. this will help you. There was a great Scientist Clausius, and second law of thermodynamics has one statement followed by his name called Clausius statement. We have something call Clausius inequality. Are you familiar with that?. if not, I have attached to support my arguments. The option C is correct, but for reversible process only. And they have not specified about the reversibility of process, so you couldn't tick option C as correct. that's why option D is correct according to Clausius inequality. Now in some of your comments, you were asking about option B and C are same? No, the explanation is again the same. Option B is equal to option C if and only if the process is reversible. dS=dQ/T is true for reversible process only. I think, now you have no doubt left, if no?, do let me know. Best regards.