Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
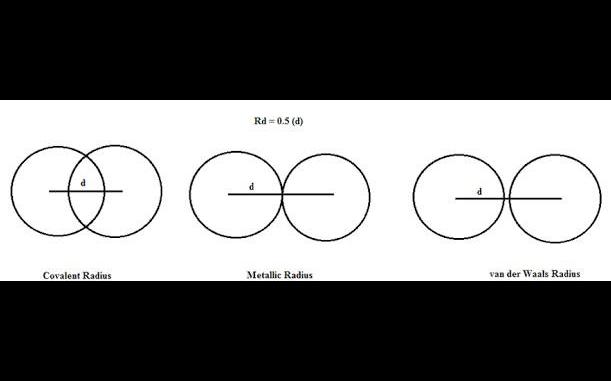
What is bragg slater radii van der waals rao rragg slater radii : bra88 and slater proposed set of internally consisten
Van der Waals rao Rragg Slater Radii : Bra88 and Slater proposed set of internally consistenta atomic radi." ,These added, irrespective of the bond type involved in the interaction between two atoms t egood correlations with observed internuclear distances. In general terms, the Bragg Slater radi epresent the radii of inner cores of electrons, which in turn determine how closely two atomic specdies approach each other. The Bragg's Slater radii of elements of second period are given ahead molecules are those in which the shared electron pairs are equally attracted by the bonded al
- 0 Likes
- 7 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Lingareddy 1
![best-answer]()
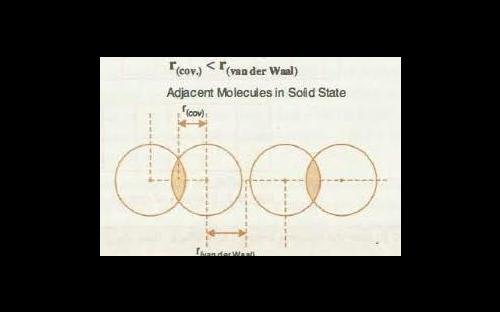
Van der waals radius is greater than covalent radius because van der waals radius is the radius between two different molecules whereas covalent radius is the radius between two different atoms.
-
![comment-profile-img]() >
>
Lingareddy 1
vanderwall radius :- The Van der Waals radius is equal to one half the distance between two unbonded atoms when the electrostatic forces between them are balanced. In other words, it is half of the closest distance between two atoms that aren't bonded or within the same molecule.
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Suresh khokhar Best Answer
https://books.google.co.in/books?id=be0T64ZWp9EC&pg=PA96&lpg=PA96&dq=bragg+slater&source=bl&ots=5QzCrqFvoI&sig=ACfU3U2MpOSWhhMBx3fIRZ-mVJ3LSW7nzQ&hl=en&sa=X&ved=2ahUKEwiXxc2GpqvpAhUj73MBHSz9ALQQ6AEwAXoECAIQAQ#v=onepage&q=bragg%20slater&f=false.
-
![comment-profile-img]() >
>
Suresh khokhar
what's is your question
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
 >
>


 >
>







Dinesh khalmaniya 1![best-answer]()
Another explanation of bragg slater radii