Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
What is the use of copper salt in a grignard reagent
what is the use of copper salt in a Grignard reagent
- 0 Likes
- 2 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Dinesh khalmaniya 1 Best Answer
An organocopper can be made by transmetallating a grignard reagent. This also acts as a nucleophilillic carbanion: Copper is less electropositive, so it produces a less-charged (or "softer") carbanion. And we know soft nucleophiles are a good way to encourage conjugate addition instead of direct attack. Example:
![conjugate addition of organocopper.png]()
![eduncle-logo-app]()
sir I only get that it's made grignard reagent a soft nucleophile
![eduncle-logo-app]()
so that grignard reagent will attack on less polarizes site
![eduncle-logo-app]()
here due to Cu+ ion nature of RMgX changed to soft so it will give micheal addition. in the question if you see RMgX/CUX the Michele addition product is formed. if only RMgX is given the direct addition
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
 >
>
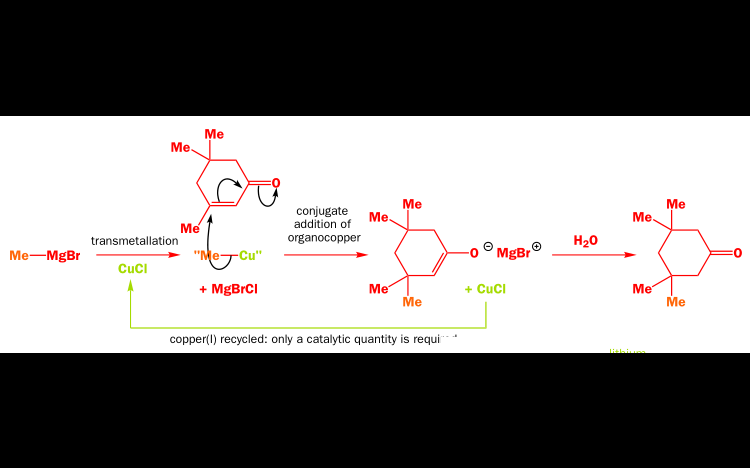








Krishna parmar![best-answer]()
Since the discovery that copper(I) halides catalyze the conjugate addition of Grignard reagents in 1941, organocopper reagents have emerged as weakly basic, nucleophilic reagents for substitution and addition reactions. ... Organocopper complexes (RCu) are produced when a copper(I) halide and organolithium are combined.