Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Sumaita Molla posted an Question
- IIT JAM
- Chemistry (CY)
Which one is more basic between aniline &pyridine ,,explain
which one is more basic between Aniline &Pyridine ,,explain
- 0 Likes
- 4 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
Amol ashok pawar Best Answer
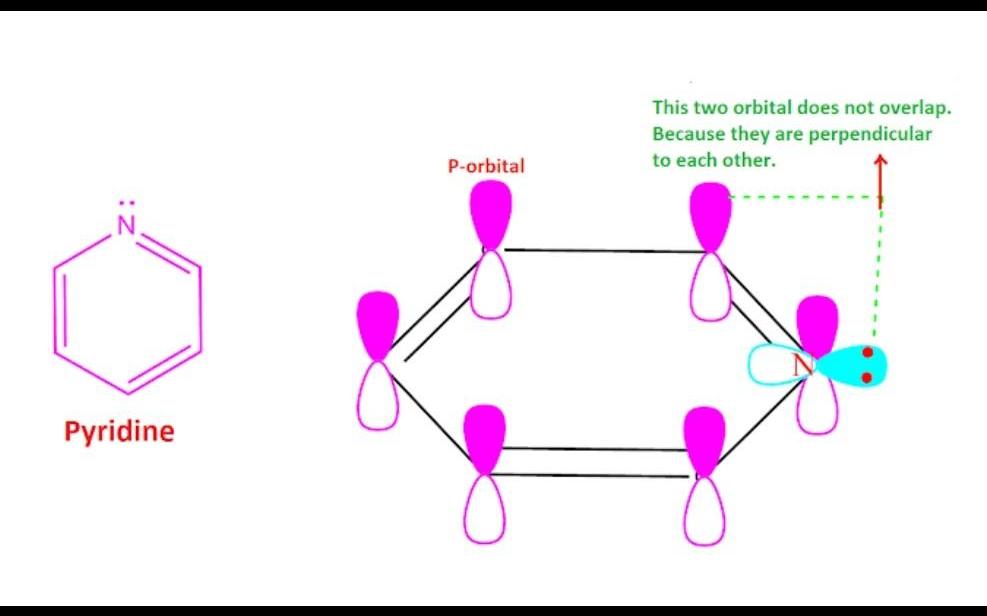
The strength of basicity depends on the tendency to donate an electron to the electron deficient element, ion or group. In case of aniline, the lone pair of electrons on nitrogen atom takes part in the resonance with the pi electron of benzene ring like structure. As a result, the availability of electrons for donation is not easy which decrease the tendency to donate electron and hence basicity of aniline decreases. On the other hand, in case of pyridine the lone of electron on nitrogen atom does not participate in the resonance with the pi electron of pyridine ring . Because, the p-orbital of carbon atom is mutually perpendicular to the p-orbital of nitrogen atom. Therefore, the resonance between p-orbital of carbon atom and p-orbital of nitrogen atom does not occur. Hence ,the lone pair of electron on ‘N’-atom is available for donation to the electron deficient element, ion or group.Consequently, pyridine is more basic than aniline.
![cropped5030593735822962225.jpg]()
![cropped635987770799423901.jpg]()
![eduncle-logo-app]()
I've got my answer,,Thank u sir
-
![comment-profile-img]() >
>
Priyanshu kumar
pyridine is more basic
![eduncle-logo-app]()
The strength of basicity depends on the tendency to donate an electron to the electron deficient element, ion or group. In case of aniline, the lone pair of electrons on nitrogen atom takes part in the resonance with the pi electron of benzene ring. ... Consequently, pyridine is more basic than aniline.
![eduncle-logo-app]()
got this?
![eduncle-logo-app]()
yes
![eduncle-logo-app]()
Why the LP on N atom in pyridine doesn't undergo resonance?
![eduncle-logo-app]()
because it is localised here....sp2 hybridized nitrogen atom..also pyridine is aromatic
![eduncle-logo-app]()
please mark the answer if you got this
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>



 >
>







Suman Kumar
https://iitjamchemistrysolvedpapers.blogspot.com/