Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
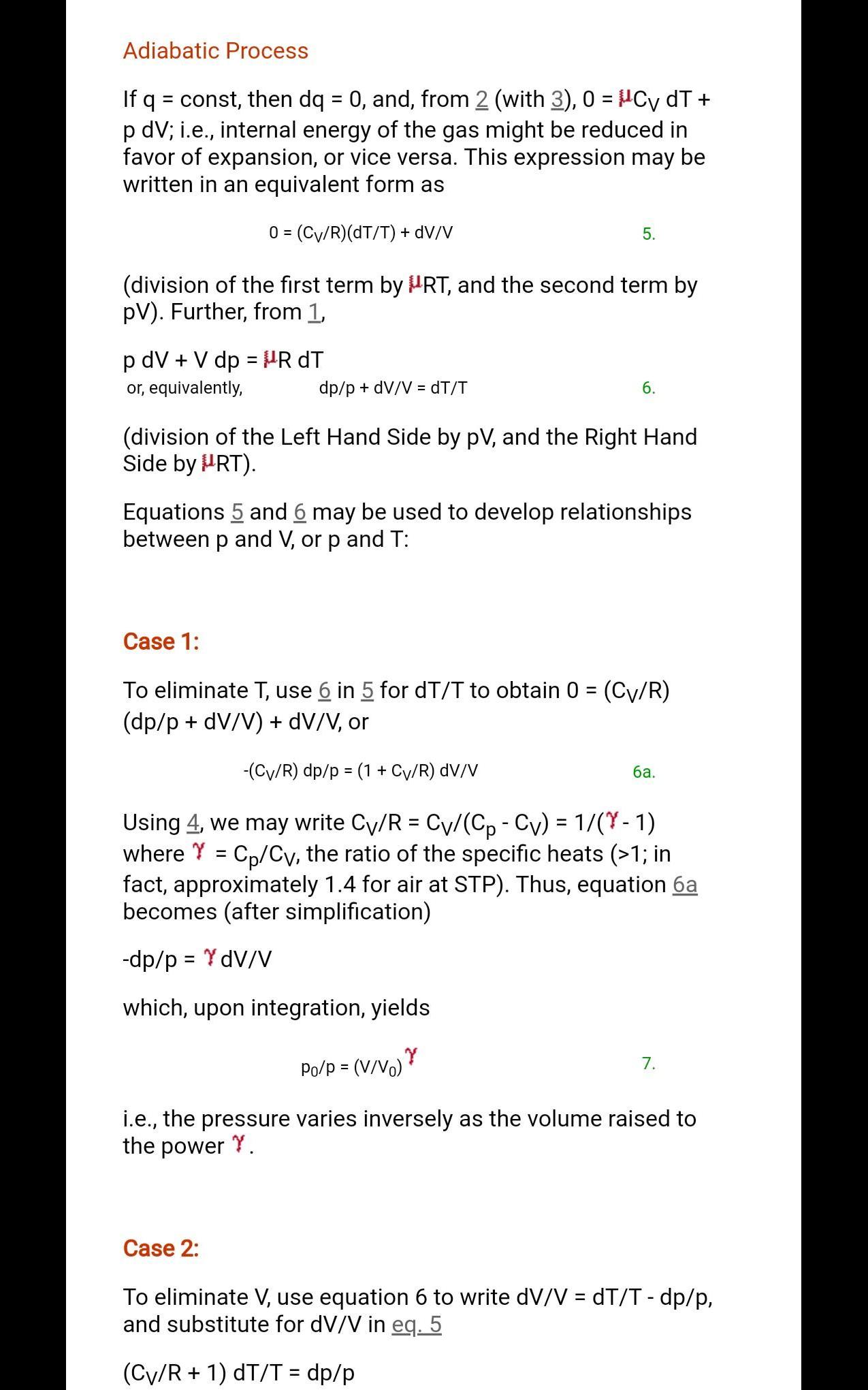
Why adiabetic is occur at constant volume only plz tell...
why adiabetic is occur at constant volume only plz tell...
- 0 Likes
- 10 Comments
- 0 Shares
-
Suman Kumar
The adiabatic process i.e. the entropy of a system remains constant. In other words, a reversible adiabatic process is an isentropic process at the same time. In this irreversible adiabatic process dq = 0, but ds > 0, hence, an irreversible adiabatic process is not isentropic.
-
Suman Kumar
Every thing will be zero if volume is constant. See the formula
-
![comment-profile-img]() >
>
-
Suman Kumar
Sweta so Adibatic process cannot occur with constant volume.
-
Suman Kumar
![best-answer]()
Never adiabatic can't occur at constant volume. Remember in reversible adiabatic problem PV gamma = constant. For adiabatic process du = dw du = Cvdt There will be change in the temperature. Change in temperature must cause change in volume. Volume will cause change in pressure according to P1V1 ^gamma = P2V2 ^gamma.
-
![comment-profile-img]() >
>
Lingareddy 1
Now, since the process is adiabatic, so the heat transfer is zero and so the entropy change is zero through heat transfer. And if the process is also reversible, then there is no entropy generated inside the system and the entropy change due to entropy generation is also zero. In this way, the entropy change is zero for an adiabatic process which is also reversible.
-
![comment-profile-img]() >
>
Lingareddy 1
Entropy remains constant in an adiabatic process which is also reversible. For the sake of simplicity consider the case of closed system, i.e. a control mass, which does not exchange any mass with the surroundings. There are two ways in which the entropy of such a system can change, namely, through heat transfer at the system boundary and through entropy generation inside the system.
-
![comment-profile-img]() >
>
Lingareddy 1
adiabatic process is a thermodynamic process during which no energy is transferred as heat across the boundaries of the system. As there is no exchange of heat with surroundings, so total heat of the system remains constant.
-
![comment-profile-img]() >
>
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>


 >
>








Suman Kumar
Sweta don't be confused with linga Reddy. Entropy do remain constant but not volume. See these two formula.
For reversible Adibatic expansion increase in entropy due to volume is compensated by decrease in temperature hence entropy remain constant.
Sweta trust me your every doubts will be cleared in this picture.
Sweta please respond