Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
Why emf decreases with increase in temperature..here plz check the question first then solution..... k) + aq cacl:5/2 h,0 (sat.) + 2ag) is 0-6753 v at 25°c
Example 2. The EMF of the ell Ca,CáCl%.2-5H,O(saturated) |1 AgCl), Ag in wbich the cel ection s Ca)+ 2AgCK) + aq CaCl:5/2 H,0 (sat.) + 2Ag) is 0-6753 V at 25°C and -6915 V at O°C. Calculate the free energy change (AG), enthalpy change (A) and entropy change (A5) of tbe cell reaction at 25°C. Solution : 1. Free Energy Change (a6); : The cell reaction, requires 2 faradays of electriciy for its completiom since n=2 Free energy change is given by the relation -AG nFE (= V C) AG-2 mol x 96,485 C mol" x0-6753 VV =- 130,3329J = - 130-33 kJ Enthalpy Cbange (AF) : The EMF of the cell is given by (Eq. 4) In this case, EMF decreases with increase in temperature, i.e., (©ElGIp is negative. Thus, --6915 V-0-6753V 25 K 0-00065 V K at atmospheric pressure 0-6753 2molx96,485 C mol mol-T+ 298 K (-0-00065 V K-) aH 16717 joules=- 167-72 kJ 3. Entropy Change (A5) The eatropy change is related to enthalpy change and free energy change by the well known thermodynamc expression, AG = AH -7AS. -AS AG-AH . -130-331 lo-232 - 01238 K 128J K 298 K
- 0 Likes
- 3 Comments
- 0 Shares
-
Achyut ranjan gogoi Best Answer
see according to nernst equation we can tell that if the temperature is increase emf decreases but in this question it has given ∆e/∆t that means how emf is variate with temperature. this also depends on entropy if entropy is positive then emf is increase with temp and if entropy is negative then emf has indirectly relationship with temperature. this is not an fixed value it depends on questions and thr value given. hope this will answer ur question.
![eduncle-logo-app]()
thanku so much sir
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-












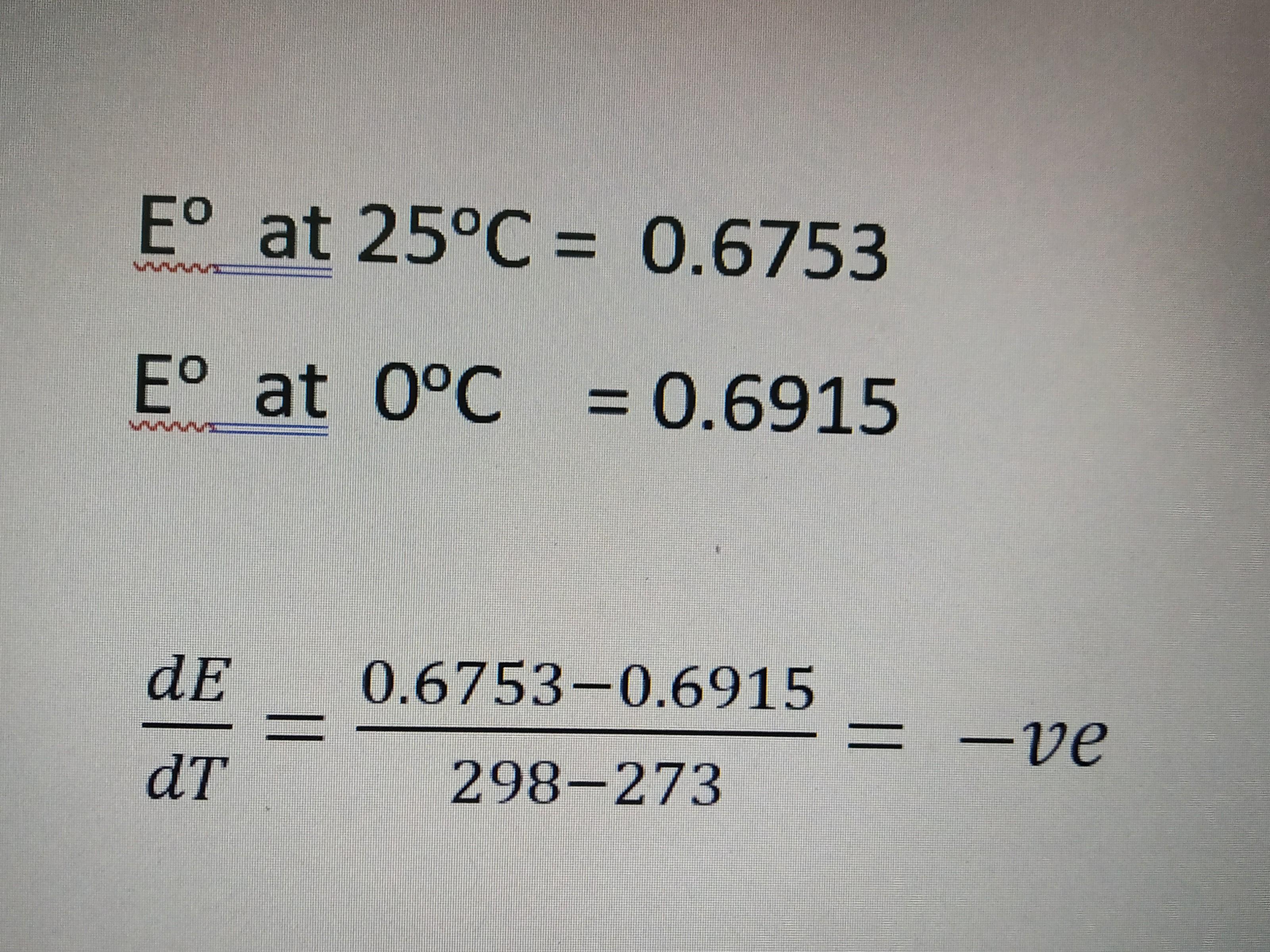







Achyut ranjan gogoi
this relationship is thermodynamic behaviour with emf.