Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Simun Mishra posted an Question
- IIT JAM
- Chemistry (CY)
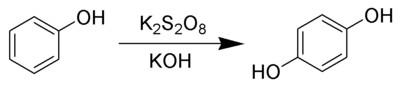
Why in presence of k2s2o8 in phenol the hydroxylation occqure in only para position ?
- 0 Likes
- 4 Comments
- 0 Shares
-
![comment-profile-img]() >
>
![eduncle-logo-app]()
check the mechanism. here phenoxide ion tautomerised to give a stable intermediate Carbanion. str 2
![eduncle-logo-app]()
sir their is some proble I can't get any reply from ur side
![eduncle-logo-app]()
now got it?
![eduncle-logo-app]()
I m checking sir
![eduncle-logo-app]()
okay
![eduncle-logo-app]()
but sir in the 2nd 2 the - charge on para why don't go for further resonance
![eduncle-logo-app]()
Simun, negative charge para pr jada stable hoga
![eduncle-logo-app]()
yha pr extended resonance he
![eduncle-logo-app]()
or para position pr negative charge ka repulsion minimum hota he so most stable so para product bnega
![eduncle-logo-app]()
extended resonance means
![eduncle-logo-app]()
sir e density ortho or pare do no me jyada hota he na
![eduncle-logo-app]()
yes
![eduncle-logo-app]()
tho fir ye after resonance para position bala negative charge kun nahi gaya ortho pe
![eduncle-logo-app]()
show nhi kiya, kyoki less stable hoga, ortho pr negative charge jada repulsion krega
![eduncle-logo-app]()
jb 1 se jada double bonds conjunction me hote he to ise extended conjunction bol dete he
![eduncle-logo-app]()
sir ortho pe kyun nahi
![eduncle-logo-app]()
less stable ortho pe kyun ho ga sir udhar bhi e density hota he na
![eduncle-logo-app]()
negative charge oxygen k lone pair se repulsion krega na
![eduncle-logo-app]()
sir but ye c tak ane se pahele B mein negative charge pe tho ata he na
![eduncle-logo-app]()
yes, it's correct 👍
![eduncle-logo-app]()
mechanism me direct de diya
![eduncle-logo-app]()
but sir iskbad bhi ek steep hoga na jisme ortho pe negative ho ga para in point of view of Stability of negative charge the reagent attack on the negative on para
![eduncle-logo-app]()
is this statement correct sir
![eduncle-logo-app]()
yes, it show 5 RS pr yha only para product bnega so sari resonating structure show ni krke, only para wali show kr di
![eduncle-logo-app]()
so only para is stable here
![eduncle-logo-app]()
yes
![eduncle-logo-app]()
tq sir 😊🙏🙏
![eduncle-logo-app]()
😊
-
![comment-profile-img]() >
>
Dinesh khalmaniya 1
A reaction mechanism has been postulated which accounts for the observed para substitution featuring the tautomeric para carbanion of the starting phenolate ion:[5] It begins with nucleophilic displacement on the peroxide oxygen of the peroxodisulfate (peroxydisulfate) ion, to give an intermediate sulfate group (3), which is then hydrolyzed to the hydroxyl group. The reaction is disadvantaged by low chemical yields with recovery of starting material and complete consumption of the persulfate. It is suggested that the phenol in many cases is a catalyst converting the persulfate into a sulfate.
![cropped184927028964658586.jpg]()
-
![comment-profile-img]() >
>
Priyanshu kumar
see simun elbs persulfate oxidation mechanism https://youtu.be/MJx3DKfstbE
![eduncle-logo-app]()
Simun here para products predominates and when there is any substituent present in para position then it gives ortho product..... see the intermediate
![eduncle-logo-app]()
got this simun??
![eduncle-logo-app]()
please respond simun
![eduncle-logo-app]()
sry sir
![eduncle-logo-app]()
why only para
![eduncle-logo-app]()
here simun stabilised carbanion is formed
![eduncle-logo-app]()
and also.it is experimentally evidenced that para is major product
![eduncle-logo-app]()
ok sir tqsir
![eduncle-logo-app]()
welcome Simun😊
![eduncle-logo-app]()
got this na??
![eduncle-logo-app]()
please accept the answer if you got this simun😊🙏
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>


 >
>







Simun mishra
can u pls explain me about this