Time management is very much important in IIT JAM. The eduncle test series for IIT JAM Mathematical Statistics helped me a lot in this portion. I am very thankful to the test series I bought from eduncle.
Nilanjan Bhowmick AIR 3, CSIR NET (Earth Science)Shweta Thakur posted an Question
- IIT JAM
- Chemistry (CY)
Why this is reversible reaction with carboxylic acids, in presence of a few drops h,so or dry hcl gas as catalyst, to
in an organiC compound. 3. Reaction with carboxylic acids. Alcohols react with carboxylic acids, in presence of a few drops H,SO or dry HCl gas as catalyst, to form esters. For example, Conc. HSO RCO-OH +H-OR RCO-OR+H,O Acid Alcohol Ester O O IOOO Conc.HsO4 P.8 CH-C OH + HOCH,CH CH-C-OCH,CH3 +H,0 oldo Ethanoic acid Ethanol Ethyl ethanoate his reaction which is slow and reversihle* is_called esterification. When HCl gas is used a
- 0 Likes
- 2 Comments
- 0 Shares
-
![comment-profile-img]() >
>
-
![comment-profile-img]() >
>
Dinesh khalmaniya 1
![best-answer]()
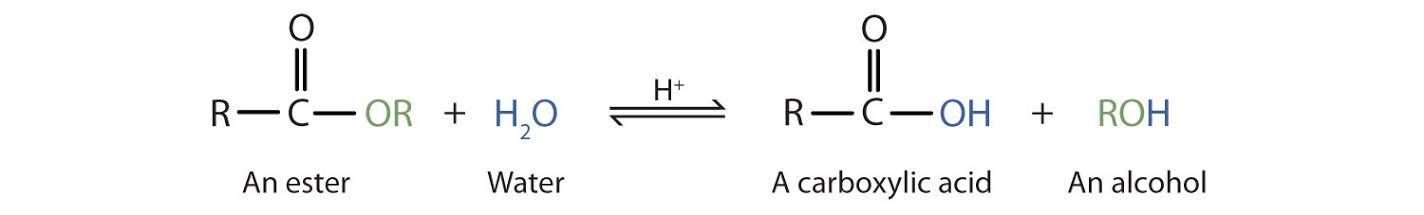
Because on hydrolysis of ester in presence of Acid gives alcohol and acid. Acidic hydrolysis is simply the reverse of esterification. The ester is heated with a large excess of water containing a strong-acid catalyst. ... As a specific example, butyl acetate and water react to form acetic acid and 1-butanol. The reaction is reversible and does not go to completion
![images (15).jpeg]()
Do You Want Better RANK in Your Exam?
Start Your Preparations with Eduncle’s FREE Study Material
- Updated Syllabus, Paper Pattern & Full Exam Details
- Sample Theory of Most Important Topic
- Model Test Paper with Detailed Solutions
- Last 5 Years Question Papers & Answers
Sign Up to Download FREE Study Material Worth Rs. 500/-










 >
>
 >
>








Priyanshu kumar Best Answer
Acidic hydrolysis is simply the reverse of esterification. The ester is heated with a large excess of water containing a strong-acid catalyst. Like esterification, the reaction is reversible and does not go to completion.
sir
sir
sir